Organic Chemistry
- • Amides (2600)
- • Alcohols, Phenols, Phenol Alcohols (305)
- • Nitrogen Compounds (248)
- • Nitrile Compound (83)
- • Hydrazine or Hydroxylamine Derivatives (203)
- • Quinones (38)
- • Ethers and Derivatives (182)
- • Aldehydes (108)
- • Carboxylic Acids and Derivatives (754)
- • Hydrocarbons and Derivatives (2305)
- • Ketones (71)
- • Inorganic Acid Esters (169)
- • Heterocyclic Ring (332)
- • Organometallic Compounds (958)
- • Organosulfur Compounds (69)
- • Phosphines (725)
- • Organometalate (283)
- • Organic Fluorine Compound (2103)
- • Triazenes (18)
- • Semicarbazides (33)
- • Organoselenium Compounds (28)
- • Nitro Compounds (94)
- • Nitrates (15)
- • Lactones (240)
- • Imines (24)
- • Free Radicals (46)
- • Dioxins and Dioxin-like Compounds (31)
- • Cyanates (31)
- • Coordination Complexes (3059)
- • Boron Compounds (84)
- • Azides (66)
- • Arsenicals (56)
Related News
Sort Hydrocarbons and Derivatives Alphabetically
Hydrocarbons and Derivatives
Get Hydrocarbons and Derivatives Raw Materials by Region-
Industrial Grade / 99%
-
Industrial Grade / 99.5%
$90/MT FOB
-
Pharmacy Grade / 99%
-
![Cyclopropane buy Cyclopropane]()
Request for quotation , get quotes from more suppliers.
(+)-Epichlorohydrin
(67843-74-7)-
Industrial Grade / 99%
-
Industrial Grade / 99%
-
-
Pharmaceutical Grade / 99%
Request for quotation , get quotes from more suppliers.
(-)-Epichlorohydrin
(51594-55-9)-
-
- / 99.00%
-
Pharmaceutical Grade / 99%
-
Industrial Grade / 99%
Request for quotation , get quotes from more suppliers.
o-Xylene
(95-47-6)-
Industrial Grade / 99%
$5-6.5/KG FOB
-
Industrial Grade / 99%
-
-
Pharmacy Grade / 99.9%
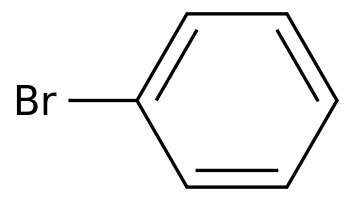
Bromobenzene
(108-86-1)-
Industrial Grade / 99%
-
Industrial Grade / 99%
-
$2-2.2/MT EXW
-
![Bromobenzene buy Bromobenzene]()
Request for quotation , get quotes from more suppliers.
-
- / 99.00%
-
Pharmacy Grade / 99%
$10/KG EXW
-
Pharmacy Grade / 99%
-
- / 99.0%
Request for quotation , get quotes from more suppliers.
Anthracene
(120-12-7)-
Industrial Grade / 99%
-
Industrial Grade / 99%
-
Chemical Grade / -
-
![Anthracene buy Anthracene]()
Request for quotation , get quotes from more suppliers.
Acetylene
(74-86-2)-
Industrial Grade / 99%
-
Industrial Grade / 99%
$2/KG FOB
-
![Acetylene buy Acetylene]()
-
![Acetylene buy Acetylene]()
Industrial Grade / 99%
Request for quotation , get quotes from more suppliers.
-
strong product / 99.5%
-
Industrial Grade / 99%
-
Industrial Grade / 99%
-
![Butane, 2,3-dimethyl- buy Butane, 2,3-dimethyl-]()
Request for quotation , get quotes from more suppliers.
Cyclopentane
(287-92-3)-
- / 99%
-
Industrial Grade / 99%
-
Industrial Grade / 99%
-
$2000-2100/MT FOB
Request for quotation , get quotes from more suppliers.
More Information
Hydrocarbons, also known as carbonaceous compounds, are organic compounds composed solely of carbon and hydrogen. These compounds, commonly referred to as hydrocarbons, form through reactions with chlorine, bromine vapor, oxygen, and other elements. Methane (CH4) is the simplest hydrocarbon molecule, consisting of a central carbon atom connected to four hydrogen atoms.
In our daily lives, we frequently encounter hydrocarbons. For instance, propane in natural gas and butane in lighters are both examples of hydrocarbons. They serve as fuels due to the vast energy stored within their covalent bonds. During combustion, this energy is released in the form of heat as they react with oxygen to produce carbon dioxide and water.
Hydrocarbons encompass various types, including:
● aromatic hydrocarbons
● saturated hydrocarbons
● aliphatic hydrocarbons