Inorganic Chemistry
- • Elementary Substance (160)
- • Industrial Gases (16)
- • Inorganic Bases (53)
- • Inorganic Acid (52)
- • Inorganic Salts (1906)
- • Oxides and Peroxides (183)
- • Silica gel (5)
- • Non-metallic Minerals (14)
- • Coating Materials (24)
- • Phosphorus Compounds (50)
- • Iron Compounds (21)
- • Bromine Compounds (20)
Related News
Sort Inorganic Salts Alphabetically
Inorganic Salts
Get Inorganic Salts Raw Materials by Region-
-
- / 98%
$1/KG EXW
-
- / 99.00%
-
Industrial Grade / 98%
Request for quotation , get quotes from more suppliers.
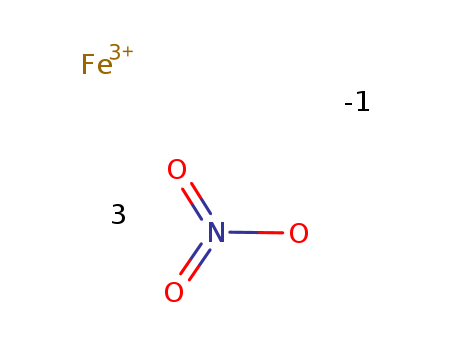
Iron nitrate (Fe(NO3)3) nonahydrate
(7782-61-8)-
Industrial Grade / 99%
$5-6.5/KG FOB
-
Chemical Grade / 99%
-
- / 99.00%
-
Industrial Grade / 99%
Request for quotation , get quotes from more suppliers.
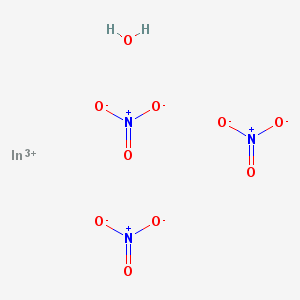
Indium trichloride
(10025-82-8)-
Chemical Grade / 98%
-
- / 98%
$1/KG EXW
-
- / 99.00%
-
- / 99%
Request for quotation , get quotes from more suppliers.
-
- / 99.00%
-
Reagent Grade / 99%
$100-110/KG FOB
-
-
Pharmacy Grade / 99%
Request for quotation , get quotes from more suppliers.
-
- / 99.00%
-
Industrial Grade / 99%
-
Industrial Grade / 99%
$2-2.1/MT FOB
-
Industrial Grade / 99.9%
$5-6/G EXW
Request for quotation , get quotes from more suppliers.
Source Inorganic Salts Products Supply
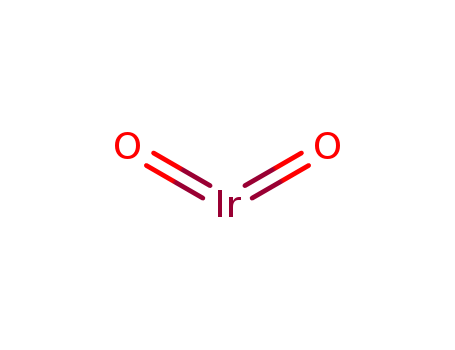
Iridium trichloride hydrate
(14996-61-3)-
Industrial Grade / 99%
-
- / 99.00%
-
industrial Grade / 98%
-
Industrial Grade / 99%
$2-2.1/MT FOB
Request for quotation , get quotes from more suppliers.
Iron sulfide (FeS)
(1317-37-9)-
- / 99.00%
-
Industrial Grade / 99%
-
Industrial Grade / 99%
$75-79/KG FOB
-
![Iron(II) sulfide (hexagonal) buy Iron(II) sulfide (hexagonal)]()
Industrial Grade / 99.0%
Request for quotation , get quotes from more suppliers.
-
- / 99.00%
-
Different Grade / 99.9%
$0.1/KG EXW
-
Industrial Grade / 99%
-
Industrial Grade / 99%
Request for quotation , get quotes from more suppliers.
-
Reagent Grade / 99%
-
- / 99.00%
-
![INDIUM NITRATE buy INDIUM NITRATE]()
-
![Indium(III) nitrate hydrate buy Indium(III) nitrate hydrate]()
- / 99%
Request for quotation , get quotes from more suppliers.
I,I′-[2,2′-Dimethyl-5,5′-bis(1-methylethyl)[1,1′-biphenyl]-4,4′-diyl] dihypoiodite
(552-22-7)-
Industrial Grade / 99%
-
![Thymol iodide buy Thymol iodide]()
-
![THYMOL IODIDE buy THYMOL IODIDE]()
-
![THYMOL IODIDE buy THYMOL IODIDE]()
Industrial Grade / 99%
Request for quotation , get quotes from more suppliers.
More Information
Inorganic salts, also known as minerals, encompass both major and trace elements, constituting essential substances in human metabolism. Most often, when referring to inorganic salts, we imply pure substances, such as laboratory-grade sodium chloride. We do not label it as table salt because additional substances are often added to table salt.
The primary distinction between inorganic and organic salts lies in the nature of their anions. Much of their chemical properties are determined by these anions. For instance, inorganic salts typically exhibit strong hydrophilicity, while organic salts may display affinity towards certain nonpolar reagents.
Despite their low concentration in cells and the human body, inorganic salts play significant roles. A diversified diet, with less animal fat consumption and more consumption of coarse grains like brown rice and corn, and limited intake of refined flour, helps maintain the normal levels of inorganic salts within the body.
Common inorganic salts include:
● sodium chloride
● potassium chloride
● calcium carbonate
● magnesium sulfate
● ammonium nitrate