3-Amino-5-(trifluoromethyl)benzonitrile
-
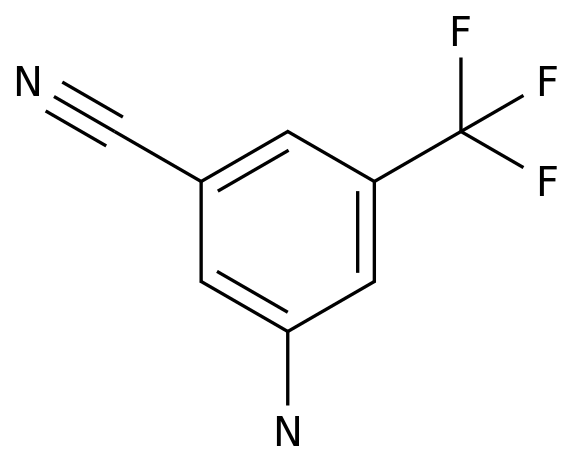
3-Amino-5-(trifluoromethyl)benzonitrile
structure -
-
CAS No:
49674-28-4
-
Formula:
C8H5F3N2
-
Chemical Name:
3-Amino-5-(trifluoromethyl)benzonitrile
-
Synonyms:
Benzonitrile,3-amino-5-(trifluoromethyl)-;3-Amino-5-(trifluoromethyl)benzonitrile;5-Amino-α,α,α-trifluoro-m-tolunitrile
- Categories:
-
CAS No:
3-Amino-5-(trifluoromethyl)benzonitrile Use and Manufacturing
To a solution of 3-nitro-5-(trifluoromethyl)benzonitrile (432.0 mg, 2.0 mmol) in ethanol (5 mL) were added zinc (1.3 mg, 20 mmol), ammonium chloride aqueous solution (5 mL). The resultant mixture was stirred for 14h at 60°C. The organic layer was filtered and the filtrate was concentrated under reduced pressure. The crude was partitioned between H3-Amino-5-trifluoromethyl-benzonitrile.; To a solution of 3-nitro-5-(trifluoromethyl)benzonitrile (500 mg, 2.31 mmol) and acetic acid (6.62 mL, 116 mmol) in EtOH (10 mL) and water (10 mL) is added tin (II) chloride (2.61 g, 11.6 mmol) and the reaction mixture is heated at 80 C for 6 h. The solvent is removed in vacuo and then the residue is portioned between DCM and 4 N HCl solution. The organic layer separated and then dried over anhydrous NaDMF (10 mL) was deoxygenated by bubbling with nitrogen for 30 minutes before a solution of 3-amino-5-bromobenzotrifluoride (500 mg, 2.08 mmol) in DMF was added. The solution was bubbled with nitrogen for another 10 minutes fore zinc cyanide (147 mg, 1.25 mmol) and Tetrakis (triphenylphospine) -Pd (96 mg, 0.083 mmol) were added. The mixture was deoxygenated for another 15 minutes before heated to 80 °C in a sealed high pressure tube overnight. The reaction was diluted with ethyl acetate, washed with an ammonia hydroxide solution (2x), and concentrated IN VACUO. The crude product was purified by preparation plates (30/70 ethyl acetate/hexanes) to yield 43-1 (289 mg, 74.5percent). 1H NMR (400 MHz, CDC13) .sect. 7.25 (d, J=0.6 Hz, 1H), 7.08 (s, 1H), 7.06 (d, J=0.9 Hz, 1H). LC-MS: MW calculated 186.13, found 227.8 (M+ACETONITRILE+).Specific examples of the trifluoromethylbenzonitrile include...4-methoxy-3-trifluoromethylbenzonitrile, 2-amino 3-trifluoromethylbenzonitrile, 2-amino-5-trifluoromethylbenzonitrile, 2-amino-6-trifluoromethylbenzonitrile, 3-amino-5-trifluoromethylbenzonitrile, 4-amino-2-trifluoromethylbenzonitrile, 4-amino-3-trifluoromethylbenzonitrile, 2, 3-bis(trifluoromethyl)benzonitrile, ...Specific examples of the trifluoromethylbenzonitrile include...4-methoxy-3-trifluoromethylbenzonitrile, 2-amino-3-trifluoromethylbenzonitrile, 2-amino-5-trifluoromethylbenzonitrile, 2-amino-6-trifluoromethylbenzonitrile, 3-amino-5-trifluoromethylbenzonitrile, 4-amino-2-trifluoromethylbenzonitrile, 4-amino-3-trifluoromethylbenzonitrile, 2, 3-bis(trifluoromethyl)benzonitrile, ...A solution of ethyl 6-chloro-8-(cyclopropyl(4-methoxybenzyl)amino) imidazo[l, 2-b]pyridazine-3-carboxylate (400 mg, 1.0 mmol) in dimethylacetamide (DMA, 4 mL) was purged with nitrogen for 10 minutes at which point 3-amino-5- (trifluoromethyl)benzonitrile (371 mg, 2.00 mmol), Pd2dba3 (91 mg, 0.10 mmol), Xantphos (289 mg, 0.50 mmol), cesium carbonate (1300 mg, 3.99 mmol) and copper(I) iodide (38.0 mg, 0.200 mmol) were added. The tube was sealed and then heated to 125 °C, after one hour the reaction was filtered through celite, eluting with ethyl acetate and the ethyl acetate was washed with brine, dried over sodium sulfate, filtered and concentrated. The crude product was then purified using automated chromatography. The collected fractions were concentrated and then suspended in dichloromethane (4 mL), triethylsilane (1.59 mL, 9.98 mmol) and trifluoroacetic acid (TFA, 2 mL) were then added and the reaction was stirred for 30 minutes. The solvent was removed in vacuo and the crude product was purified using automated chromatography (25-30percent ethyl acetate:petroleum ether) to provide ethyl 6-(3-cyano-5-(trifluoromethyl)phenylamino)-8- (cyclopropylamino)imidazo[l, 2-b]pyridazine-3-carboxylate (200 mg, 47percent yield). NMR (400MHz, DMSO-de) delta 9.96 (s, 1H), 8.81 (s, 1H), 8.45 (s, 1H), 8.04 (s, 1H), 7.88 (s, 1H), 7.79 (s, 1H), 6.22 (s, 1H), 4.41 (q, J=7.0 Hz, 2H), 2.59 (m, 1H), 1.36 (t, J=7.0 Hz, 3H), 0.84 (m, 2H), 0.70 (m, 2H).To a solution of
Computed Properties
Molecular Weight:186.13
XLogP3:1.8
Hydrogen Bond Donor Count:1
Hydrogen Bond Acceptor Count:5
Exact Mass:186.04048265
Monoisotopic Mass:186.04048265
Topological Polar Surface Area:49.8
Heavy Atom Count:13
Complexity:227
Covalently-Bonded Unit Count:1
Compound Is Canonicalized:Yes
Recommended Suppliers of 3-Amino-5-(trifluoromethyl)benzonitrile
-
![China]() CN
CN
4 YRS
Business licensedTrader Supplier of Intermediates,Building blocks,API,Silicones,Peptides,Lab chemicals,Biochemicals,Pharmaceuticals,Screening Compounds,Food Additives
Learn More Other Chemicals
-
3-AMINO-5-(TRIFLUOROMETHYL)BENZONITRILE
30825-34-4
-
4-(Trifluoromethyl)benzeneacetic acid
32857-62-8
-
3-BROMO-2-FLUORO-6-PICOLINE
375368-78-8
-
2,4-Diisopropylphenol Formula
2934-05-6
-
1,2-Diiodotetrafluoroethane Formula
354-65-4
-
6-METHYL-2-PIPERIDINECARBOXYLICACID Formula
99571-58-1
-
2,4-Dichloro-6-picoline Structure
42779-56-6
-
1-BROMO-1H,1H,2H,2H-PERFLUORODECANE Structure
21652-57-3
-
What is Perfluorohexanoic acid
307-24-4
-
What is 6-chloro-2-naphthalenethiol
392330-26-6
3-Amino-5-(trifluoromethyl)benzonitrile
SDSRequest for Quotation





