3-AMINO-4-FLUOROPHENYLBORONIC ACID
-
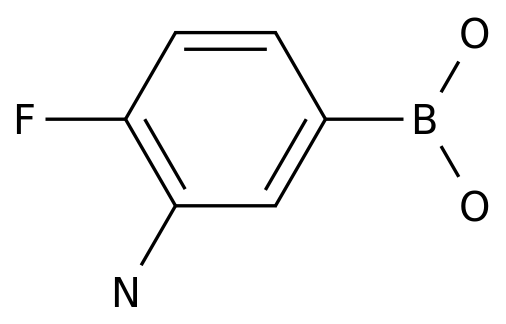
3-AMINO-4-FLUOROPHENYLBORONIC ACID
structure -
-
CAS No:
873566-75-7
-
Formula:
C6H7BFNO2
-
Chemical Name:
3-AMINO-4-FLUOROPHENYLBORONIC ACID
-
Synonyms:
3-Amino-4-fluorobenzeneboronic acid;5-Borono-2-fluoroaniline;Boronicacid, (3-amino-4-fluorophenyl)- (9CI);(3-aMino-4-fluorophenyl)boronic acid hydrochloride
- Categories:
-
CAS No:
Characteristics
66.5
-0.33110
White to off-white Solid
1.3±0.1 g/cm3
345.974°C at 760 mmHg
163.0±30.7 °C
1.555
Safety Information
22
Xi,Xn
P261, P264, P270, P271, P280, P301+P312, P302+P352, P304+P340, P305+P351+P338, P312, P321, P330, P332+P313, P337+P313, P362, P403+P233, P405, P501
H302
|Warning|H302 (66.67%): Harmful if swallowed [Warning Acute toxicity, oral]|P261, P264, P270, P271, P280, P301+P312, P302+P352, P304+P340, P305+P351+P338, P312, P321, P330, P332+P313, P337+P313, P362, P403+P233, P405, and P501|Aggregated GHS information provided by 3 companies from 3 notifications to the ECHA C&L Inventory. Each notification may be associated with multiple companies.
3-AMINO-4-FLUOROPHENYLBORONIC ACID Use and Manufacturing
Hydrogenation of 3-nitro-4-fluorobenzeneboronic acid (63 g) was placed in a 500 mL single-necked flask. Methanol (100 mL) and palladium carbon (6.3 g, 10percent) were added and sealed with a stopper. Water chestnut vacuum, needle into the hydrogen. The reaction flask was placed in an oil bath at 55 ° C and the reaction was stirred. Hydrogen balloon reaction after the replacement until no longer absorb hydrogen. The reaction solution was filtered through a Buchner funnel to remove palladium-carbon, dried to dryness, beaten with n-heptane, and dried in vacuo. The solid was dissolved in methanol (30 mL), 150 mL of water was added, a large amount of solid was precipitated, the methanol was distilled off and a portion of water was distilled off and the temperature was lowered to 5 ° C. The Buchner funnel was filtered off as a pale gray solid and dried in vacuo to give The final product was 42.6 g, the yield was 80.7percent, the structure was confirmed by 1Hz NMR, and the HPLC was 99.1percent.98percent of 2-fluoro-5-bromonitrobenzene (211 g, 0.96 mol) and iron powder (212 g, 3.8 mol) were mixed, 1200 ml of ethanol was added and glacial acetic acid (456 g, 7.6 mol) was added dropwise at room temperature. The reaction solution was treated with an oil bath at 60 ° CThe reaction solution was completed by a gas phase controlled reaction. The reaction solution was filtered through a Buchner funnel pad and the filtrate was evaporated to dryness and extracted with ethyl acetateN-heptane (volume ratio 1: 4) was purified by column and evaporated to give pure product 122.5 g, 98percent GC, yield 62.5percentIn a reaction vessel, 5-[4-chloro-3-(trifluoromethoxy)phenyl]-3, 6-dihydro-2H-1, 3, 4- oxadiazin-2-one (105 mg, 356 muiotaetaomicronIota, Intermediate 73), In a reaction vessel, 5-[4-chloro-3-(trifluoromethyl)phenyl]-3, 6-dihydro-2H-1, 3, 4- oxadiazin-2-one (90.0 mg, 323 muiotaetaomicronIota, Intermediate 64),
Computed Properties
Molecular Weight:154.94
Hydrogen Bond Donor Count:3
Hydrogen Bond Acceptor Count:4
Rotatable Bond Count:1
Exact Mass:155.0553868
Monoisotopic Mass:155.0553868
Topological Polar Surface Area:66.5
Heavy Atom Count:11
Complexity:136
Covalently-Bonded Unit Count:1
Compound Is Canonicalized:Yes
Recommended Suppliers of 3-AMINO-4-FLUOROPHENYLBORONIC ACID
-
![China]() CN
CN
3 YRS
Business licensedTrader Supplier of 1H,1H,2H,2H-PERFLUORODECYLTRIMETHOXYSILANEInquiryCAS No.: 873566-75-7Content: 99% -
![China]() CN
CN
4 YRS
Business licensed Certified factoryManufactory Supplier of Custom Synthetic Chemicals,Biological Stains,Pharmaceutical Intermediates,Cosmetic Grade Chemicals,Enzyme,Nucleosides,Indicators,Speciality Chemicals,Biological Chemicals,Enzyme Substrates -
![China]() CN
CN
1 YR
Business licensed Certified factoryManufactory Supplier of 4-(Pentafluorothio)aniline,Pentafluoro,Pentafluorothio,Pentafluorosulfur,[4-(bromomethyl)phenyl]-pentafluoro-lambda6-sulfane,[4-(pentafluoro-sulfanyl)phenyl]acetic acidInquiryCAS No.: 873566-75-7Grade: Industrial GradeContent: 95%
Learn More Other Chemicals
-
(4-BROMOPHENYL)(4-BOC-1,4-DIAZEPAN-1-YL)ACETIC ACID
834884-94-5
-
2-(4-CHLOROPHENYL)-3-[(4-METHYLPHENYL)SULFANYL]-4-QUINOLINECARBOXYLIC ACID
477867-85-9
-
3-[(2-chloroanilino)carbonyl]-7-oxabicyclo[2.2.1]heptane-2-carboxylic acid
1026678-38-5
-
CIS-4-(4-CYANOBENZOYL)CYCLOHEXANE-1-CARBOXYLIC ACID Formula
735269-97-3
-
2-chloro-5-[5-({3-ethyl-2-[(4-methoxyphenyl)imino]-4-oxo-1,3-thiazolidin-5-ylidene}methyl)-2-furyl]benzoic acid Formula
443875-50-1
-
(2-{[1-(4-chlorophenyl)ethylidene]hydrazono}-4-oxo-1,3-thiazolidin-5-yl)acetic acid Formula
311785-26-9
-
5-[(6-chloro-1,3-benzothiazol-2-yl)amino]-5-oxopentanoic acid Structure
444937-77-3
-
5-[(5-chloro-2-methoxybenzyl)amino]-2-(4-morpholinyl)benzoic acid Structure
765925-15-3
-
What is 4-[({4-[(6-chloro-3-pyridinyl)methoxy]-3-methoxybenzyl}amino)methyl]benzoic acid
880809-62-1
-
What is 4-{[(5-chloro-2-ethoxyphenyl)sulfonyl]amino}benzoic acid
927637-85-2
3-AMINO-4-FLUOROPHENYLBORONIC ACID
SDSRequest for Quotation





