Eslicarbazepine acetate
-
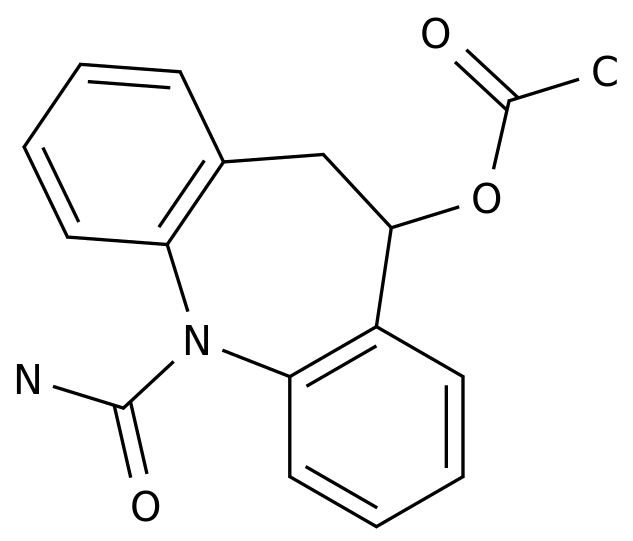
Eslicarbazepine acetate
structure -
-
CAS No:
236395-14-5
-
Formula:
C17H16N2O3
-
Chemical Name:
Eslicarbazepine acetate
-
Synonyms:
5H-Dibenz[b,f]azepine-5-carboxamide,10-(acetyloxy)-10,11-dihydro-,(10S)-;(10S)-10-(Acetyloxy)-10,11-dihydro-5H-dibenz[b,f]azepine-5-carboxamide;BIA 2-093;(S)-(-)-10-Acetoxy-10,11-dihydro-5H-dibenz[b,f]azepine-5-carboxamide;(S)-10-Acetoxy-10,11-dihydro-5H-dibenz[b,f]azepine-5-carboxamide;Eslicarbazepine acetate;Sep 0002093;Exelief;(S)-(-)-10-Acetoxy-10,11-dihydro-5H-dibenzo[b,f]azepine-5-carboxamide;Aptiom;(S)-5-Carbamoyl-10,11-dihydro-5H-dibenzo[b,f]azepin-10-yl acetate;Zebinix;Exalief
- Categories:
-
CAS No:
Description
Eslicarbazepine acetate, an antiepileptic drug, is a dual a dual Inhibitor of β-Secretase and voltage-gated sodium channel.
Eslicarbazepine acetate is the acetate ester, with S configuration, of licarbazepine. An anticonvulsant, it is approved for use in Europe and the United States as an adjunctive therapy for epilepsy. It has a role as an anticonvulsant and a drug allergen. It is an acetate ester, a dibenzoazepine, a carboxamide and a member of ureas. It derives from a licarbazepine.|Eslicarbazepine acetate (ESL) is an anticonvulsant medication approved for use in Europe, the United States and Canada as an adjunctive therapy for partial-onset seizures that are not adequately controlled with conventional therapy. Eslicarbazepine acetate is a prodrug that is rapidly converted to eslicarbazepine, the primary active metabolite in the body. Eslicarbazepine's mechanism of action is not well understood, but it is known that it does exert anticonvulsant activity by inhibiting repeated neuronal firing and stabilizing the inactivated state of voltage-gated sodium channels, thus preventing their return to the activated state during which seizure activity can occur. Eslicarbazepine acetate is marketed as Aptiom in North America and Zebinix or Exalief in Europe. It is available in 200, 400, 600, or 800mg tablets that are taken once daily, with or without food. Eslicarbazepine acetate is associated with numerous side effects including dizziness, drowsiness, nausea, vomiting, diarrhea, headache, aphasia, lack of concentration, psychomotor retardation, speech disturbances, ataxia, depression and hyponatremia. It is recommended that patients taking eslicarbazepine acetate be monitored for suicidality.
Eslicarbazepine acetate Basic Attributes
296.324
296.32
1592732-453-0
BEA68ZVB2K
DTXSID90178308
N03AF04
Characteristics
72.6
2
white solid
1.3±0.1 g/cm3
186-187 °C
427.4°C at 760 mmHg
212.3±31.5 °C
1.655
Water solubility of eslicarbazepine acetate is low at less than 1 mg/mL including at different pH values. Its main metabolite eslicarbazepine has a greater water solubility of 4.2 mg/mL.
2-8°C
Safety Information
NONH for all modes of transport
3
36/37/38
26-36
Xi
P261-P305 + P351 + P338
H315-H319-H335
|Warning|H315 (98.91%): Causes skin irritation [Warning Skin corrosion/irritation]|P261, P264, P271, P280, P302+P352, P304+P340, P305+P351+P338, P312, P321, P332+P313, P337+P313, P362, P403+P233, P405, and P501|Aggregated GHS information provided by 92 companies from 2 notifications to the ECHA C&L Inventory. Each notification may be associated with multiple companies.
Toxicity
There are no adequate and well-controlled studies of the use of eslicarbazepine acetate in pregnant women. In studies conducted in pregnant mice, rats, and rabbits, eslicarbazepine acetate did show developmental toxicities, including teratogenicity, embryolethality, and fetal growth retardation, at clinically relevant doses. Drug-induced liver injury ranging from mild to moderate elevations in transaminases (>3 times the upper limit of normal) to rare cases with concomitant elevations of total bilirubin (>2 times the upper limit of normal) have been reported with the use of eslicarbazepine. Overdose with eslicarbazepine acetate appears similar to its known adverse reactions and includes symptoms of hyponatremia, dizziness, nausea, vomiting, somnolence, euphoria, oral paraesthesias, ataxia, and diplopia. There is no specific antidote for eslicarbazepine acetate overdose and it should be treated primarily with supportive measures. If required, the drug may be removed by gastric lavage, partially by hemodialysis or inactivated with activated charcoal.
Eslicarbazepine is bound to plasma proteins at a relatively low rate of <40%, independent of concentration. In vitro studies have shown that plasma protein binding is not relevantly affected by the presence of other medications such as warfarin, diazepam, digoxin, phenytoin or tolbutamide. Similarly, the binding of these medications was not significantly affected by the presence of eslicarbazepine.
Drug Information
Eslicarbazepine acetate is indicated as adjunctive therapy in the treatment of partial-onset seizures that are not adequately controlled with conventional therapy in epileptic patients.|FDA Label|Zebinix is indicated as adjunctive therapy in adults, adolescents and children aged above 6 years, with partial-onset seizures with or without secondary generalisation.|Exalief is indicated as adjunctive therapy in adults with partial-onset seizures with or without secondary generalisation.|Treatment of epilepsy with partial-onset seizures
Eslicarbazepine acetate is associated with a dose- and concentration-dependant increase in heart rate and prolongation of PR interval.
Drugs used to prevent SEIZURES or reduce their severity. (See all compounds classified as Anticonvulsants.)|A class of drugs that inhibit the activation of VOLTAGE-GATED SODIUM CHANNELS. (See all compounds classified as Voltage-Gated Sodium Channel Blockers.)
Eslicarbazepine active metabolite has a high bioavailability and reaches peak serum concentration 1-4 hours after a given dose. Eslicarbazepine acetate absorption is not affected by food.|Eslicarbazepine acetate and its metabolites are eliminated primarily via renal excretion. Eslicarbazepine active metabolite is excreted two-thirds in the unchanged form and one-third as a glucuronide conjugate. This accounts for around 90% of total metabolites excreted, with the remaining 10% being minor metabolites. Renal tubular reabsorption is expected to occur with eslicarbazepine.|The apparent volume of distribution of eslicarbazepine is 61.3 L for a body weight of 70 kg based on population PK analysis.|Renal clearance of eslicarbazepine was found to be approximately 20 mL/min in healthy subjects with normal renal function.
Eslicarbazepine acetate is rapidly and extensively metabolized to its major active metabolite, eslicarbazepine, via hydrolytic first-pass metabolism. Eslicarbazepine corresponds to about 92% of systemic exposure. Minor active metabolites (R)-licarbazepine and oxcarbazepine consist of <5% of systemic exposure. Active metabolites are then metabolized to inactive glucuronides that correspond to about 3% of systemic exposure. Eslicarbazepine had a moderate inhibitory effect on CYP2C19 and a mild activation of UGT1A1-mediated glucuronidation when studied in human hepatic microsomes. It has been shown to induce CYP3A4 enzymes in vivo.
The apparent plasma half-life of eslicarbazepine is 10-20 hours in healthy subjects and 13-20 hours in epilepsy patients. Steady-state plasma concentrations are attained after 4 to 5 days of once daily dosing.
Eslicarbazepine acetate is converted to the active metabolite eslicarbazepine which carries out its anticonvulsant activity. The exact mechanism of action is unknown, but it is thought to involve the inhibition of voltage-gated sodium channels. In in vitro electrophysiological studies, eslicarbazepine was shown to inhibit repeated neuronal firing by stabilizing the inactivated state of voltage-gated sodium channels and preventing their return to the activated state. In vitro studies also showed eslicarbazepine inhibiting T-type calcium channels, which likely also has a role in anticonvulsant activity.
10-acetoxy-10,11-dihydro-5H-dibenz(b,f)azepine-5-carboxamide
Eslicarbazepine acetate Use and Manufacturing
Eslicarbazepine acetate, (BIA 2-093), is a promising antiepileptic drug structurally related to Carbamazepine and Oxcarbazepine.
Human drugs -> Zebinix -> EMA Drug Category|Antiepileptics -> Human pharmacotherapeutic group|Human drugs -> Exalief -> EMA Drug Category|Human Drugs -> EU pediatric investigation plans|Human Drugs -> FDA Approved Drug Products with Therapeutic Equivalence Evaluations (Orange Book) -> Active Ingredients
Computed Properties
Molecular Weight:296.32
XLogP3:2
Hydrogen Bond Donor Count:1
Hydrogen Bond Acceptor Count:3
Rotatable Bond Count:2
Exact Mass:296.11609238
Monoisotopic Mass:296.11609238
Topological Polar Surface Area:72.6
Heavy Atom Count:22
Complexity:440
Defined Atom Stereocenter Count:1
Covalently-Bonded Unit Count:1
Compound Is Canonicalized:Yes
Recommended Suppliers of Eslicarbazepine acetate
-
![China]() CN
CN
4 YRS
Business licensed Certified factoryManufactory Supplier of Agrochemicals,Daily Chemicals,Catalysts & Chemical Auxiliary Agents,Extract,Inorganic Chemicals,Organic Intermediate,Pigment & Dyestuff,Polymer,Flavour & Fragrance,Basic Organic Chemicals,Pharmaceutical -
![China]() CN
CN
4 YRS
Business licensed Certified factoryManufactory Supplier of Custom Synthetic Chemicals,Biological Stains,Pharmaceutical Intermediates,Cosmetic Grade Chemicals,Enzyme,Nucleosides,Indicators,Speciality Chemicals,Biological Chemicals,Enzyme Substrates -
![China]() CN
CN
2 YRS
Business licensedTrader Supplier of Ethyl 6,4-Dihydroxybenzaldehyde,tetramethylpyrazine,Spermine,Lipoic Acid,Aripiprazole,3,Edoxaban,Rutin,Tetrahydrocurcumin,Pregabalin Powder,Olopatadine Hydrochloride,β-Nicotinamide Mononucleotide,Gabapentin powder,8-dichlorooctanoate,Meloxicam,Febuxostat,(R)-3-Piperidinamine dihydrochloride,Azilsartan,Voriconazole,Magnolol,Paclitaxel Powder,Ezetimibe,Monobenzone Powder,(R)-3-(Boc-Amino)piperidine,Quercetin
Learn More Other Chemicals
-
3-[4-(3,4-dichlorophenyl)-1,3-thiazol-2-yl]-2-oxo-2H-chromen-7-yl acetate
333319-28-1
-
4-[2-({2,4-dichloro-6-nitrophenoxy}acetyl)carbohydrazonoyl]-2-methoxyphenyl acetate
507244-69-1
-
[(5R,7R,9R,10S,11R,13S,17R)-11-hydroxy-17-[(2S,5R)-2-hydroxy-5-(2-methylpropyl)-2,5-dihydrofuran-3-yl]-4,4,8,10,13-pentamethyl-3-oxo-5,6,7,9,11,12,16,17-octahydrocyclopenta[a]phenanthren-7-yl] acetate
101508-37-6
-
(2R)-14-[(E)-{(7aR)-1-[(2R,3E,5R)-5,6-Dimethyl-3-hepten-2-yl]-7a- methyloctahydro-4H-inden-4-ylidene}methyl]-7,12-dioxo-1,2,3,4,5,7 ,12,14-octahydrophthalazino[2,3-b]phthalazin-2-yl acetate Formula
169737-23-9
-
4-(3-(3-(2-chlorophenyl)-3-oxopropanoyl)-2-hydroxy-4,6-diMethoxyphenyl)-1-Methylpiperidin-3-yl acetate Formula
244136-25-2
-
(Z,Z)-7,11-Hexadecadienyl acetate Formula
52207-99-5
-
(Z)-9-Hexadecenyl acetate Structure
34010-20-3
-
Picrasin B acetate Structure
30315-04-9
-
What is [2-[(8S,9S,10R,13S,14S,17R)-11,17-dihydroxy-10,13-dimethyl-3-oxo-2,6,7,8,9,11,12,14,15,16-decahydro-1H-cyclopenta[a]phenanthren-17-yl]-2-oxoethyl] acetate
53187-58-9
-
What is [2-[(6S,8S,9S,10R,11S,13S,14S,17R)-11,17-dihydroxy-6,10,13-trimethyl-3-oxo-2,6,7,8,9,11,12,14,15,16-decahydro-1H-cyclopenta[a]phenanthren-17-yl]-2-oxoethyl] acetate
1625-11-2





