2-Chloro-4-methylbenzaldehyde
-
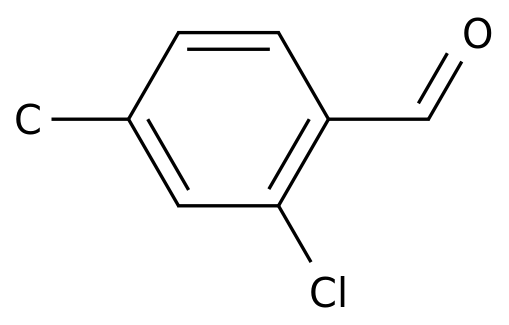
2-Chloro-4-methylbenzaldehyde
structure -
-
CAS No:
50817-80-6
-
Formula:
C8H7ClO
-
Chemical Name:
2-Chloro-4-methylbenzaldehyde
-
Synonyms:
2-CHLORO-4-METHYLBENZALDEHYDE;3-Chloro-4-formyltoluene, 2-Chloro-p-tolualdehyde;2-Chloro-p-tolualdehyde;Benzaldehyde,2-chloro-4-methyl-
- Categories:
-
CAS No:
Safety Information
NONH for all modes of transport
3
22-37/38-41-51
26-39-45
Xn
P261-P280-P305 + P351 + P338
H302-H315-H318-H335
|Danger|H302 (100%): Harmful if swallowed [Warning Acute toxicity, oral]|P261, P264, P270, P271, P280, P301+P312, P302+P352, P304+P340, P305+P351+P338, P310, P312, P321, P330, P332+P313, P362, P403+P233, P405, and P501|Aggregated GHS information provided by 39 companies from 2 notifications to the ECHA C&L Inventory. Each notification may be associated with multiple companies.
2-Chloro-4-methylbenzaldehyde Use and Manufacturing
165 ml (413 mmol) of 2.5 M nBuLi in hexanes was added dropwise over 1 h to a solution of 82.2 g (400 mmol) of 4-bromo-3-chlorotoluene in 400 ml of THF cooled to -88 °C. The resulting mixture was stirred for 30 min at this temperature, and then 44.0 g (602 mmol) of DMF was added dropwise by vigorous stirring for 10 min. The reaction mixture was stirred overnight at room temperature, and then 100 ml of water and 400 ml of 3 N HC1 were added at 0 °C. The organic layer was separated, the aqueous layer was extracted with 2 x 125 ml of dichloromethane. The combined organic extract was dried over K2CO3 and then passed through a short layer of silica gel 60 (40-63 μιη). The silica gel layer was additionally washed by 50 ml of dichloromethane. The combined organic eluate was evaporated to dryness to give a slightly orange liquid which was then distilled under vacuum to give 58.0 g (94percent) of the title product (b.p. 99-102 °C/11 mm Hg, ) as a colorless liquid crystallized overnight at room temperature.165 ml ( 413 mmol) of 2.5 M nBuLi in hexanes was added dropwise over 1 hto a solution of 82.2 g ( 400 mmol) of 3-chloro-4-bromo-toluene in 400 ml of THFcooled to -88°C. The resulting mixture was stirred for 30 min at this temperature, and then 44.0 g (602 mmol) ofDMF was added dropwise over 10 min by vigorousstirring. The reaction mixture was stirred overnight at room temperature, then cooled to ooc in an ice bath and then 100 ml of water and 400 ml of 3N HCl were added.The organic layer was separated and the aqueous layer was extracted with 2x 125 mlof dichloromethane. The combined organic extract was dried over K2C03 and thenpassed through a short layer of silica gel 60 ( 40-63 )liD). The silica gel layer wasadditionally washed with 50 ml of dichloromethane. The combined organic elute was evaporated to dryness to give a slightly orange liquid which was then distilled invacuum to give 58.0 g (94percent) ofthe title product (b.p. 99-102°C/11 mm Hg, ) as acolorless liquid that crystallized overnight at room temperature. Anal. calc. for C8H7Cl0: C, 62.15; H, 4.56. Found: C, 62.24; H, 4.45.1H NMR (CDCh): b 10.4 (s, 1H, CHO), 7.80 (d, J= 7.8 Hz, 1H, 6-H), 7.25(s, 1H, 3-H), 7.17 (d, J = 7.8 Hz, 1H, 5-H), 2.40 (s, 3H, 4-Me).
Computed Properties
Molecular Weight:154.59
XLogP3:2.4
Hydrogen Bond Acceptor Count:1
Rotatable Bond Count:1
Exact Mass:154.0185425
Monoisotopic Mass:154.0185425
Topological Polar Surface Area:17.1
Heavy Atom Count:10
Complexity:124
Covalently-Bonded Unit Count:1
Compound Is Canonicalized:Yes
Recommended Suppliers of 2-Chloro-4-methylbenzaldehyde
-
![China]() CN
CN
4 YRS
Business licensedTrader Supplier of Intermediates,Building blocks,API,Silicones,Peptides,Lab chemicals,Biochemicals,Pharmaceuticals,Screening Compounds,Food Additives -
![China]() CN
CN
3 YRS
Business licensedTrader Supplier of 1H,1H,2H,2H-PERFLUORODECYLTRIMETHOXYSILANE
Learn More Other Chemicals
-
4-Mercaptophenylboronic acid
237429-33-3
-
(4-BENZYLOXYCARBONYLPHENYL)BORONIC ACID
184000-11-1
-
(4-chlorophenyl)methoxy-trimethylsilane
14856-74-7
-
4,4'-DIBROMO-TRANS-STILBENE Formula
18869-30-2
-
(R)-(-)-1-[(S)-2-(DI(3,5-DIMETHYL-4-METHOXYPHENYL)PHOSPHINO)FERROCENYL]ETHYLDICYCLOHEXYLPHOSPHINE Formula
360048-63-1
-
4-BENZYLOXY-3-CHLOROPHENYLBORONIC ACID Formula
845551-44-2
-
4,5-dichloro-4,5-difluoro-2,2-bis(trifluoromethyl)-1,3-dioxolane Structure
60644-92-0
-
2-Methyl-2H-indazole-4-boronic acid pinacol ester Structure
885698-95-3
-
What is 3-HEPTAFLUOROPROPYL-5-METHYL-4-NITROPYRAZOLE
82633-69-0
-
What is 2,6-Dinitro-4-(trifluoromethyl)phenol
393-77-1





