2-Deoxy-D-ribono-1,4-lactone
-
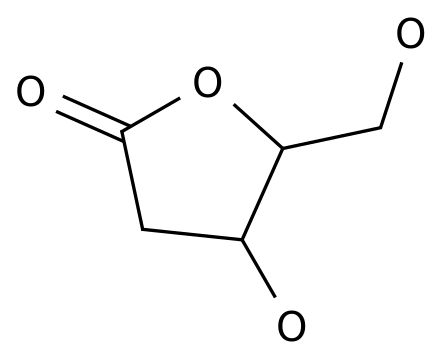
2-Deoxy-D-ribono-1,4-lactone
structure -
-
CAS No:
34371-14-7
-
Formula:
C5H8O4
-
Chemical Name:
2-Deoxy-D-ribono-1,4-lactone
-
Synonyms:
D-erythro-Pentonic acid,2-deoxy-,γ-lactone;2-Deoxy-D-erythro-pentono-γ-lactone;2-Deoxy-D-ribono-1,4-lactone;2-Deoxy-D-ribonolactone;2′-Deoxyribolactone;(4S,5R)-4-Hydroxy-5-hydroxymethylfuran-2-one;2-Deoxyribonolactone;819081-95-3
- Categories:
-
CAS No:
Description
2-deoxy-D-ribono-1,4-lactone is a ribonolactone. It has a role as a metabolite. It derives from a D-ribonic acid.
2-Deoxy-D-ribono-1,4-lactone Use and Manufacturing
The 2-deoxyribose 16 (1.03 g, 7.45 mmol) was added to a solution of Br2-Deoxyribono-lactone (2)To a solution of 2-deoxy-D-ribose (42.0 g, 313 mmol) in 800 mL of water was added Br2 (42 mL). The flask was sealed, and the contents were stirred at room temperature for 5 days. The resulting mixture was neutralized by adding silver carbonate until the pH was 7. The mixture was filtered and washed with water. After removal of water, the crude product was filtered through silica gel pad and eluted with ethyl acetate/MeOH (10:1 to 4:1). The filtrate was concentrated under reduced pressure to yield 2-deoxyribono-lactone 2 as a colorless gum (31.1 g, 75percent). ‘H NMR (DMSO-d6, 400 MHz) ö(ppm): 2.17 (dd, J = 17.8 and 2.4 Hz, 1H), 2.76 (dd, J = 17.8 and 6.4 Hz, 1H), 3.48-3.54 (m, 2H), 4.20-4.24 (m, 2H), 5.06 (t, J = 5.4 Hz, 1H), 5.50 (d, J = 4.0 Hz, 1H).Deoxy-D-ribose (400.0 g, 2.98 mol) was dissolved in water (1 .6 kg) under nitrogen and the solution cooled to 3 - 7 °C. Bromine (800 g, 10.0 mol, 3.36 eq.) was added at 3 - 7 °C while stirring over a period of approximately 2 hours and the stirring was continued at 3 - 7 °C for approximately 1 hour. The reaction mixture was gently warmed to 20 - 25°C and then stirred for approximately 20 hours. The reaction mixture was cooled to -5 to -7°C and a solution of sodium hydroxide (27.65percent, 720 g, 1 .67 eq.) was added while keeping the reaction temperature at -3 to -7 °C. The temperature was then adjusted to 0 - 5 °C and aqueous sodium hydroxide (9percent, 470 g, 1 .06 mol, 0.35 eq. was added at 0 - 5 °C to obtain a final pH = 1 .40. The water was distilled off at reduced pressure using a scrubber (cooled, 14percent sodium hydroxide, 0.9 L), finally at p < 5 mbar and 50 °C. In order to remove residual water from the product, 2-propanol was added portion wise to the residue followed by azeotropic distillation at reduced pressure. The final water content was determined by KF titration to be less than 1 percent. 2-Propanol (400 mL) was added to the residue and the mixture followed by filtration. The filter cake was washed with 2-propanol (1 L). The solvent was distilled off at reduced pressure. Toluene (400 mL) was added and distillation was resumed in order to remove residual 2- propanol and possibly more water. A residue of 474.6g (120percent yield) was obtained
Computed Properties
Molecular Weight:132.11
XLogP3:-0.9
Hydrogen Bond Donor Count:2
Hydrogen Bond Acceptor Count:4
Rotatable Bond Count:1
Exact Mass:132.04225873
Monoisotopic Mass:132.04225873
Topological Polar Surface Area:66.8
Heavy Atom Count:9
Complexity:122
Defined Atom Stereocenter Count:2
Covalently-Bonded Unit Count:1
Compound Is Canonicalized:Yes
Recommended Suppliers of 2-Deoxy-D-ribono-1,4-lactone
-
![China]() CN
CN
4 YRS
Business licensedTrader Supplier of Intermediates,Building blocks,API,Silicones,Peptides,Lab chemicals,Biochemicals,Pharmaceuticals,Screening Compounds,Food Additives
Learn More Other Chemicals
-
5-Bromo-2-methoxythiazole
446287-05-4
-
4-[(6,7-Dimethoxyquinolin-4-yl)oxy]aniline
190728-25-7
-
N-[3-Fluoro-4-[(methylamino)carbonyl]phenyl]-2-met
1332524-01-2
-
Glycine Formula
56-40-6
-
Sodium iodide Formula
7681-82-5
-
Bromo-2-pyridinylzinc Formula
218777-23-2
-
ethyl 2-oxo-2-(2-oxopyrrolidin-3-yl)acetate Structure
90090-56-5
-
2-Butyl-3-(4-(3-(dibutylamino)propoxy)benzoyl)-5-nitrobenzofuran Structure
141645-23-0
-
What is Macluraxanthone
5848-14-6
-
What is 2-(2-chlorophenyl)-5-[3-[5-(2-chlorophenyl)-1,3,4-thiadiazol-2-yl]phenyl]-1,3,4-thiadiazole
90032-82-9





