2,6-DICHLOROPHENYLBORONIC ACID
-
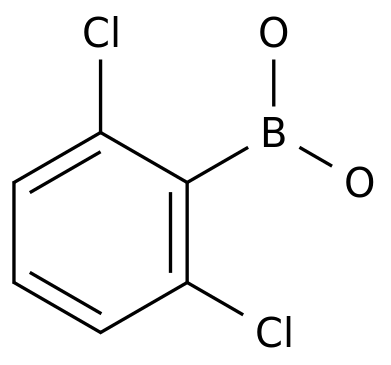
2,6-DICHLOROPHENYLBORONIC ACID
structure -
-
CAS No:
73852-17-2
-
Formula:
C6H5BCl2O2
-
Chemical Name:
2,6-DICHLOROPHENYLBORONIC ACID
-
Synonyms:
RARECHEM AH PB 0089;2,6-DICHLOROPHENYLBORONIC ACID;2,6-DICHLOROBENZENEBORONIC ACID;AKOS BRN-0086;2,6-Dichlorophenylboronic;3,6-DICHLOROPHENYLBORONICACID;2,6-Dichlorophenylboronic Acid (contains varying amounts of Anhydride);2,6-Dichlorophenylboronic acid,98%
- Categories:
-
CAS No:
2,6-DICHLOROPHENYLBORONIC ACID Basic Attributes
190.82
189.975967
4802506
-0
DTXSID30370223
29319090
Characteristics
40.5
0.67320
Off-white Powder
1.5±0.1 g/cm3
153 °C
341.8°C at 760 mmHg
160.5±30.7 °C
1.577
Insoluble in water.
Safety Information
IRRITANT
36/37/38-22-36
26-36-37/39
Xi,Xn
Irritant
P261, P264, P271, P280, P302+P352, P304+P340, P305+P351+P338, P312, P321, P332+P313, P337+P313, P362, P403+P233, P405, P501
H315
|Warning|H315 (22.45%): Causes skin irritation [Warning Skin corrosion/irritation]|P261, P264, P271, P280, P302+P352, P304+P340, P305+P351+P338, P312, P321, P332+P313, P337+P313, P362, P403+P233, P405, and P501|Aggregated GHS information provided by 49 companies from 7 notifications to the ECHA C&L Inventory. Each notification may be associated with multiple companies.
2,6-DICHLOROPHENYLBORONIC ACID Use and Manufacturing
(1) Lithiation: under the protection of nitrogen, First in a 1000mL three-necked flask, Add 50g of 1, 3-dichlorobenzene and cool to -80~-60°C.Then add 176 mL of n-butyl lithium dropwise.The dropping process is maintained at -80 to -50 ° C.After the completion of the dropwise addition, the reaction was kept for 2 hours.(2) Lithium-hydrogen exchange:117 g of tributyl borate liquid was added dropwise to the solution of the above-obtained phenyl lithium, After the addition is completed, the heat retention reaction is carried out.After the reaction, the reaction system was stirred by adding hydrochloric acid having a mass concentration of 10percent.Then let go of the layering, Extracted, Combine the organic layers, Desolvent, Hydrating succinol to obtain 51.7 g of crude 4-phenoxybenzeneboronic acid in a yield of 80percent;(3) Purification:51.7 g of crude 4-phenoxyphenylboronic acid was added to a 1 L three-necked flask.Add 200 mL of methyl tert-butyl ether to dissolve, Desolvent, Cooling, crystallization, and centrifugation gave 48.4 g of 4-phenoxybenzeneboronic acid product in a yield of 75percent.
suzuki reaction
Computed Properties
Molecular Weight:190.82
Hydrogen Bond Donor Count:2
Hydrogen Bond Acceptor Count:2
Rotatable Bond Count:1
Exact Mass:189.9759650
Monoisotopic Mass:189.9759650
Topological Polar Surface Area:40.5
Heavy Atom Count:11
Complexity:124
Covalently-Bonded Unit Count:1
Compound Is Canonicalized:Yes
Recommended Suppliers of 2,6-DICHLOROPHENYLBORONIC ACID
-
![China]() CN
CN
4 YRS
Business licensedTrader Supplier of Intermediates,Building blocks,API,Silicones,Peptides,Lab chemicals,Biochemicals,Pharmaceuticals,Screening Compounds,Food Additives
Learn More Other Chemicals
-
(4-BROMOPHENYL)(4-BOC-1,4-DIAZEPAN-1-YL)ACETIC ACID
834884-94-5
-
2-(4-CHLOROPHENYL)-3-[(4-METHYLPHENYL)SULFANYL]-4-QUINOLINECARBOXYLIC ACID
477867-85-9
-
3-[(2-chloroanilino)carbonyl]-7-oxabicyclo[2.2.1]heptane-2-carboxylic acid
1026678-38-5
-
CIS-4-(4-CYANOBENZOYL)CYCLOHEXANE-1-CARBOXYLIC ACID Formula
735269-97-3
-
2-chloro-5-[5-({3-ethyl-2-[(4-methoxyphenyl)imino]-4-oxo-1,3-thiazolidin-5-ylidene}methyl)-2-furyl]benzoic acid Formula
443875-50-1
-
(2-{[1-(4-chlorophenyl)ethylidene]hydrazono}-4-oxo-1,3-thiazolidin-5-yl)acetic acid Formula
311785-26-9
-
5-[(6-chloro-1,3-benzothiazol-2-yl)amino]-5-oxopentanoic acid Structure
444937-77-3
-
5-[(5-chloro-2-methoxybenzyl)amino]-2-(4-morpholinyl)benzoic acid Structure
765925-15-3
-
What is 4-[({4-[(6-chloro-3-pyridinyl)methoxy]-3-methoxybenzyl}amino)methyl]benzoic acid
880809-62-1
-
What is 4-{[(5-chloro-2-ethoxyphenyl)sulfonyl]amino}benzoic acid
927637-85-2





