2,6-DICHLORO-3-NITROBENZOIC ACID
-
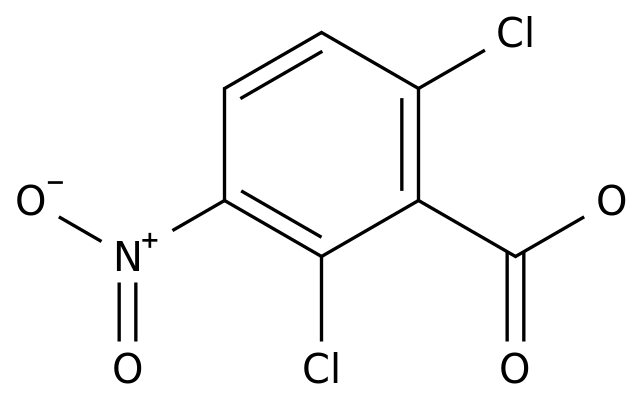
2,6-DICHLORO-3-NITROBENZOIC ACID
structure -
-
CAS No:
55775-97-8
-
Formula:
C7H3Cl2NO4
-
Chemical Name:
2,6-DICHLORO-3-NITROBENZOIC ACID
-
Synonyms:
RARECHEM AL BO 2381;TIMTEC-BB SBB003247;2,6-DICHLORO-3-NITROBENZOIC ACID;Benzoic acid, 2,6-dichloro-3-nitro-
- Categories:
-
CAS No:
Safety Information
NONH for all modes of transport
3
36/37/38
26-37/39
Xi
P261-P305 + P351 + P338
H315-H319-H335
|Warning|H315 (100%): Causes skin irritation [Warning Skin corrosion/irritation]|P261, P264, P271, P280, P302+P352, P304+P340, P305+P351+P338, P312, P321, P332+P313, P337+P313, P362, P403+P233, P405, and P501|Aggregated GHS information provided by 40 companies from 3 notifications to the ECHA C&L Inventory. Each notification may be associated with multiple companies.
2,6-DICHLORO-3-NITROBENZOIC ACID Use and Manufacturing
Step a: Synthesis of 2, 6-dichloro-3-nitro-benzoic acid2, 6-Dichloro-benzoic acid (10g, 52 mmol) was treated with a previously stirred mixture of nitric acid (15 mL) and sulfuric acid (30 mL) at 55 °C for 30 minutes followed by stirring at room temperature for 30 minutes. The reaction mixture was poured in ice cold water (1 L) and the solid obtained was filtered and dried to afford the product (9 gm). The filtrate was extracted with ethyl acetate (2 χ 200 mL). The combined organic layer was dried over anhydrous sodium sulphate and concentrated to dryness to yield more product (2 gm). (1 1 g, 88percent).(i) Step a: Synthesis of 2, 6-Dichloro-benzoic acid (10 g, 52 mmol) was treated with a previously stirred mixture of nitric acid (15 mL) and sulfuric acid (30 mL) at 55 C. for 30 minutes followed by stirring at room temperature for 30 minutes. The reaction mixture was poured in ice cold water (1 L) and the solid obtained was filtered and dried to afford the product (9 gm). The filtrate was extracted with ethyl acetate (2*200 mL). The combined organic layer was dried over anhydrous sodium sulphate and concentrated to dryness to yield more product (2 gm). (11 g, 88%). 1H NMR (400 MHz, MeOH-d4): delta 7.98 (d, J=8.8 1H), 7.67 (d, J=8.8 1H).(i) Nitrobenzoic acids may be obtained with advantage by the process according to the invention, the following being mentioned by way of example: ... 6-nitro-3-bromobenzoic acid, 3-nitro-2-chloro-4-methyl benzoic acid, 5-nitro-2-chloro-4-methyl benzoic acid, 5-nitro-2, 4-dichlorobenzoic acid,
Computed Properties
Molecular Weight:236.01
XLogP3:2.5
Hydrogen Bond Donor Count:1
Hydrogen Bond Acceptor Count:4
Rotatable Bond Count:1
Exact Mass:234.9439130
Monoisotopic Mass:234.9439130
Topological Polar Surface Area:83.1
Heavy Atom Count:14
Complexity:255
Covalently-Bonded Unit Count:1
Compound Is Canonicalized:Yes
Recommended Suppliers of 2,6-DICHLORO-3-NITROBENZOIC ACID
-
![China]() CN
CN
4 YRS
Business licensedTrader Supplier of Intermediates,Building blocks,API,Silicones,Peptides,Lab chemicals,Biochemicals,Pharmaceuticals,Screening Compounds,Food Additives -
![China]() CN
CN
3 YRS
Business licensedTrader Supplier of 1H,1H,2H,2H-PERFLUORODECYLTRIMETHOXYSILANEInquiryCAS No.: 55775-97-8Grade: Industrial GradeContent: 99%
Learn More Other Chemicals
-
(4-BROMOPHENYL)(4-BOC-1,4-DIAZEPAN-1-YL)ACETIC ACID
834884-94-5
-
2-(4-CHLOROPHENYL)-3-[(4-METHYLPHENYL)SULFANYL]-4-QUINOLINECARBOXYLIC ACID
477867-85-9
-
3-[(2-chloroanilino)carbonyl]-7-oxabicyclo[2.2.1]heptane-2-carboxylic acid
1026678-38-5
-
CIS-4-(4-CYANOBENZOYL)CYCLOHEXANE-1-CARBOXYLIC ACID Formula
735269-97-3
-
2-chloro-5-[5-({3-ethyl-2-[(4-methoxyphenyl)imino]-4-oxo-1,3-thiazolidin-5-ylidene}methyl)-2-furyl]benzoic acid Formula
443875-50-1
-
(2-{[1-(4-chlorophenyl)ethylidene]hydrazono}-4-oxo-1,3-thiazolidin-5-yl)acetic acid Formula
311785-26-9
-
5-[(6-chloro-1,3-benzothiazol-2-yl)amino]-5-oxopentanoic acid Structure
444937-77-3
-
5-[(5-chloro-2-methoxybenzyl)amino]-2-(4-morpholinyl)benzoic acid Structure
765925-15-3
-
What is 4-[({4-[(6-chloro-3-pyridinyl)methoxy]-3-methoxybenzyl}amino)methyl]benzoic acid
880809-62-1
-
What is 4-{[(5-chloro-2-ethoxyphenyl)sulfonyl]amino}benzoic acid
927637-85-2
2,6-DICHLORO-3-NITROBENZOIC ACID
SDSRequest for Quotation





