2,4-DICHLORO-6-METHYLQUINAZOLINE
-
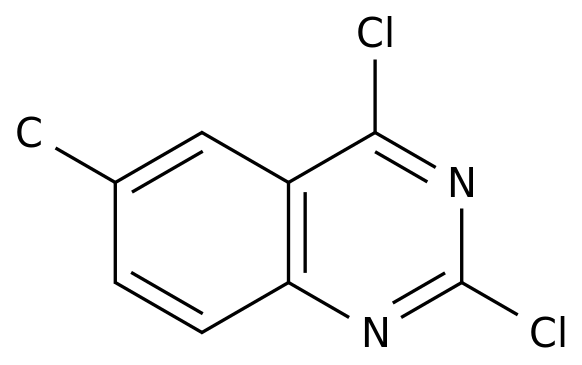
2,4-DICHLORO-6-METHYLQUINAZOLINE
structure -
-
CAS No:
39576-82-4
-
Formula:
C9H6Cl2N2
-
Chemical Name:
2,4-DICHLORO-6-METHYLQUINAZOLINE
-
Synonyms:
2,4-DICHLORO-6-METHYLQUINAZOLINE;Quinazoline, 2,4-dichloro-6-methyl-;2,4-Dichloro-6-methyl-quinazoline;2,4 Dichloro-6-methyl quinazoline;PubChem23095;SCHEMBL1993196;CTK1B3844;DTXSID60558092;METHYL19-METHYLEICOSANOATE;KS-00000I0P
- Categories:
-
CAS No:
Safety Information
P261, P264, P271, P280, P302+P352, P304+P340, P305+P351+P338, P312, P321, P332+P313, P337+P313, P362, P403+P233, P405, P501
H315
|Warning|H315 (100%): Causes skin irritation [Warning Skin corrosion/irritation]|P261, P264, P271, P280, P302+P352, P304+P340, P305+P351+P338, P312, P321, P332+P313, P337+P313, P362, P403+P233, P405, and P501|The GHS information provided by 1 company from 1 notification to the ECHA C&L Inventory.
2,4-DICHLORO-6-METHYLQUINAZOLINE Use and Manufacturing
General procedure: Quinazolin-2, 4(1H, 3H)-dione (10 g, 61.7 mmol), DIPEA (22.6 ml, 129 mmol) and POClTo a solution of 6-methyl-1 H-quinazoline-2, 4-dione (2.29 g, 0.013 mol) in 20 mL POC13 was added N, N-dimethylaniline (1.81 mL, 0. 014 mol). The mixture was heated to reflux (125 °C) and stirred for 4 hours until the starting material completely dissolved and the solution turned dark purple in color. The solution was then cooled and poured slowly on ice (40 G ; CAUTION HIGHLY EXOTHERMIC) to quench the reaction, The aqueous layer was then extracted three times with CH, CI, (40 ML). The organic layer was dried ONER HISSO4, CONCENTRATED and subjected to purification by chromatography (100percent CH2Cl2) to yield 2, 4-dichloro-6-methyl-quinazoline(2, 5 g, 90 percent) as a slightly yellow solid. P 1H NMR (400 MHZ, DMSO-D6) No. 8.05 (S, 1H), 8.01 (D, @=9.2 HZ, 1 H), 7L.94 (D, J = 8.8 HZ, 1 H), , 57 (S, 3 H).To a solution of 6-methylquinazoline-2, 4(1 H, 3H)-dione (550 mg, 3.12 mmol, ) in POCIGeneral procedure: Quinazolin-2, 4(1H, 3H)-dione (10 g, 61.7 mmol), DIPEA (22.6 ml, 129 mmol) and POCl3 (4.0 ml) were heated at reflux. After 3 hours the reaction mixture was cautiously poured over crushed ice and stirred vigorously. This aqueous mixture was extracted with CH2Cl2 DCM and the combined organic layers were washed with brine and dried over Na2SO4. Evaporation of the solvent gave a crystalline solid that was dissolved in CH2Cl2 after which it was filtered over a pad of silica using CH2Cl2 as eluent. Removal of the organic phase gave the product as 10.80 g (54.3 mmol, 88%) of a white solid.To a solution of 6-methyl-1 H-quinazoline-2, 4-dione (2.29 g, 0.013 mol) in 20 mL POC13 was added N, N-dimethylaniline (1.81 mL, 0. 014 mol). The mixture was heated to reflux (125 C) and stirred for 4 hours until the starting material completely dissolved and the solution turned dark purple in color. The solution was then cooled and poured slowly on ice (40 G ; CAUTION HIGHLY EXOTHERMIC) to quench the reaction, The aqueous layer was then extracted three times with CH, CI, (40 ML). The organic layer was dried ONER HISSO4, CONCENTRATED and subjected to purification by chromatography (100% CH2Cl2) to yield 2, 4-dichloro-6-methyl-quinazoline(2, 5 g, 90 %) as a slightly yellow solid. P 1H NMR (400 MHZ, DMSO-D6) No. 8.05 (S, 1H), 8.01 (D, @=9.2 HZ, 1 H), 7L.94 (D, J = 8.8 HZ, 1 H), , 57 (S, 3 H).To a solution of 6-methylquinazoline-2, 4(1 H, 3H)-dione (550 mg, 3.12 mmol, ) in POCI3 (4.0 mL) was added N.N'-diethylanaline (0.5 mL.). The reaction mixture was heated to 1350C and stirred for 3 h. The reaction mixture was cooled to rt and poured into ice. The solid was collected and dried to give 2, 4-dichloro-6- methylquinazoline (590 mg, 89 %), which was used for the next step without further purification.(2-CHLORO-6-METHYL-QUINAZOLIN-4-YL)- (4-METHOXY-PHENYL)-METHYL-AMINE : The above 6-methyl-quinazoline-2, 4-dione (201 mg, 1.14 mmol) and N, N- dimethylaniline (0.2 mL) were refluxed in phosphorus oxychloride (5 mL) under argon overnight. The solvent was removed by distillation under reduced pressure. The purple residue was dissolved in isopropanol (10 mL). N-METHYL-P-ANISIDINE (201 mg, 1.465 mmol) was added. The mixture was stirred at room temperature overnight. The solvent was evaporated and the residue was purified by column chromatography (SI02, EtOAc: hexanes 5-25%) to give the product as a light yellow solid (62 mg, 17 %) : H NMR (CDCI3) 7.62 (d, J = 8.7 Hz, 1H), 7.38 (dd, J = 1. 8, 8. 7 Hz, 1H), 7.16- 7.10 (M, 2H), 6.89-6. 86 (M, 2H), 6.63 (s, 1H), 3.86 (s, 3H), 3.60 (s, 3H), 2.09 (s, 3H).A suspension of 6-methyl-1H-quinazoline-2, 4-dione (1.20 g, 6.8 mmol) in POCl3 (10 mL) was heated to reflux for 6 h. After cooling to room temperature the reaction was quenched by slow, dropwise addition onto ice. The resulting solid was collected via vacuum filtration, providing the title compound.j00425j To a solution of compound B-76 (8.0 g, crude) in phosphoms oxychloride (0.24 L, 400 g, 2.6 mol) at room temperature was added dropwise N, N-dimethylaniline (5.7 mL, 5.5 g, 0.45 mol). The mixture was stirred at 120 C for 2 hours, then poured into ice-water (500 mL). The resulting solid was collected by filtration and dried in vacuum to give compound B-77 (4 g, cmde) as a yellow solid. LCMS (E): tR=0.794 mi, (ES) m/z (M+H)=2 13.0One equivalent of quinazoline-2, 4-dione and one equivalent of N, N-dimethylaniline were combined in a round bottom flask, 12 equivalents of phosphorus oxychloride was then added. The mixture was refluxed under argon until the presence of starting material was no longer seen by TLC or by LC-MS (6-24 hours). Upon completion the reaction mixture was cooled and slowly added to ice equaled to ten times that of the reaction volume. Upon precipitation the reaction was filtered and washed with water to afford the crude 2, 4-dichloroquinazoline which was purified by column chromatography using hexanes and ethyl acetate.2-Chloro-6-methylquinazolin-4(3H)-one To a solution of To a solution of General procedure: Phosphorus pentachloride (12.5g, 60.0mmol), phosphorous oxychloride (46.0mL, 502.5mmol) were added to a 250mL one-neck flask, was slowly added with stirring 6-fluoro-quinazoline -2, 4 (1H, 3H) - dione (3.6g, 20.0mmol), the reaction was gradually warmed to reflux, the reaction is stopped after 9 hours, cooled, solvent was distilled off under reduced pressure, the residue was slowly poured into 400mL ice-water mixture. after stirring for 0.5 hours, extracted with dichloromethane (250mL × 3), combined dichloromethane layer was separated and purified directly by column chromatography on silica gel (petroleum ether / ethyl acetate (v / v) = 30/1) to give the title compound (white solid, 3.735g , 86.0%).in place of 1-(3, 4-dimethoxyphenyl)-3-phenoxycarbonylurea, there is produced, respectively: (a) 2, 4-dichloro-quinazoline, (b) 2, 4-dichloro-8-methylquinazoline, (c) 2, 4-dichloro-7-methylquinazoline, (d)
Computed Properties
Molecular Weight:213.06
XLogP3:3.8
Hydrogen Bond Acceptor Count:2
Exact Mass:211.9908036
Monoisotopic Mass:211.9908036
Topological Polar Surface Area:25.8
Heavy Atom Count:13
Complexity:189
Covalently-Bonded Unit Count:1
Compound Is Canonicalized:Yes
Recommended Suppliers of 2,4-DICHLORO-6-METHYLQUINAZOLINE
-
![China]() CN
CN
1 YR
Business licensed Certified factoryManufactory Supplier of 4-(Pentafluorothio)aniline,Pentafluoro,Pentafluorothio,Pentafluorosulfur,[4-(bromomethyl)phenyl]-pentafluoro-lambda6-sulfane,[4-(pentafluoro-sulfanyl)phenyl]acetic acidInquiryCAS No.: 39576-82-4Grade: Industrial GradeContent: 95%
Learn More Other Chemicals
-
2-Bromophenacyl bromide, 90%
49851-55-0
-
1H-Indazol-7-ol
81382-46-9
-
6-Chloro-4-forMyl-nicotinic acid
1031433-06-3
-
3-Hydroxy-2,4,5-trifluorobenzoic acid Formula
116751-24-7
-
2-Methyl-1-heptene Formula
15870-10-7
-
1-(3,5-Dinitrophenyl)ethanone Formula
14401-75-3
-
α-Amino-3-bromobenzeneacetic acid Structure
79422-73-4
-
6-(BROMOMETHYL)-1,3-BENZOTHIAZOLE,97% Structure
499770-85-3
-
What is ethyl 5-hydroxy-2-Methylnicotinate
60390-47-8
-
What is 2-bromo-1-(4-bromo-3-fluorophenyl)ethanone
1003879-02-4
2,4-DICHLORO-6-METHYLQUINAZOLINE
SDSRequest for Quotation





