2-Hydroxyethyl methacrylate
-
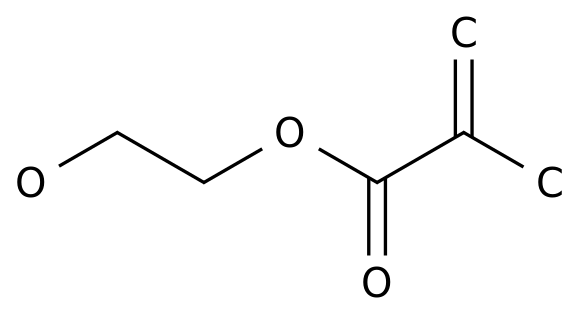
2-Hydroxyethyl methacrylate
structure -
-
CAS No:
868-77-9
-
Formula:
C6H10O3
-
Chemical Name:
2-Hydroxyethyl methacrylate
-
Synonyms:
2-Propenoic acid,2-methyl-,2-hydroxyethyl ester;Methacrylic acid,2-hydroxyethyl ester;Methacrylic acid,ester with glycol;Glycol methacrylate;Glycol monomethacrylate;Monomer MG 1;β-Hydroxyethyl methacrylate;2-Hydroxyethyl methacrylate;Ethylene glycol monomethacrylate;2-(Methacryloyloxy)ethanol;Ethylene glycol methacrylate;Acryester HO;Rocryl 400;Light Ester HO;Bisomer SR;Mhoromer BM 903;NSC 24180;Blemmer E;Acryester HISS;Mhoromer BM 905;GE 610;HEMA;Light Ester HO 250;Bisomer HEMA;HEMA 90;EB 109 (monomer);EB 109;2-HEMA;Kayarad 2-HEMA;Acryester HOMA;Light Ester HO 250M;EM 321;2-Hydroxyethyl 2-methylprop-2-enoate;Acryl Ester HO;Light Ester HO 250N;Visiomer HEMA 98;M 0085;JB 4 Plus;Visiomer HEMA 97;H 140643;51026-91-6;58308-22-8;60974-06-3;61497-49-2;112813-65-7;123991-13-9;132051-71-9;141668-69-1;151638-45-8;155280-45-8;173306-28-0;201463-85-6;203300-24-7;203497-53-4;211862-46-3;212555-08-3;219840-96-7;225107-31-3;282528-79-4;473256-73-4;1136534-55-8;1151978-80-1;1184921-46-7;1206159-39-8;1260379-87-0;1418001-98-5;1449201-78-8;2231343-05-6;2242757-54-4
- Categories:
-
CAS No:
Description
colourless liquid
Liquid|COLOURLESS LIQUID.
2-hydroxyethyl methacrylate is an enoate ester that is the monomethacryloyl derivative of ethylene glycol. It has a role as a polymerisation monomer and an allergen. It derives from an ethylene glycol and a methacrylic acid.|2-Hydroxyethyl Methacrylate is a hydroxyester compound and a resin monomer used in desensitizing dentin. By applying 2-hydroxyethyl methacrylate locally to sensitive teeth, sensitive areas in the teeth get sealed and block the dentinal tubules at the dentin surface from stimuli that cause pain. This prevents excitation of the tooth nerve and relieves pain caused by tooth hypersensitivity.
2-Hydroxyethyl methacrylate Basic Attributes
130.14200
130.14
212-782-2
6E1I4IV47V
1724
24180
DTXSID7022128
C47791
Clear mobile liquid
2916140000
Characteristics
46.53000
0.5
Liquid
1.034 g/cm3 @ Temp: 25 °C
-12ºC
67 °C @ Press: 3.5 Torr
64.043ºC
1.441
Solubility in water: miscible
2-8ºC
0.01 mm Hg ( 25 °C)
5 (vs air)
LD50 orally in Rabbit: 5050 mg/kg LD50 dermal Rabbit > 3000 mg/kg
Heat of polymerization: 49.8 kJ/mole
Safety Information
NONH for all modes of transport
1
R36/38; R43
S26-S28A
OZ4725000
Xi
Store only if stabilized. Keep in the dark. Cool. Ventilation along the floor.
Unstable - may polymerize in the absence of stabilizer. May be stabilized with, or contain small amounts of, diethylene glycol monomethacrylate, di(ethylene glycol)dimethacrylate, methacrylic acid. Incompatible with strong oxidizing agents, free radical initiators, peroxides, steel. Closed containers may explode if heated due to runaway polymer
P280-P305 + P351 + P338-P333 + P313-P337 + P313
H315-H317-H319
SRP: At the time of review, criteria for land treatment or burial (sanitary landfill) disposal practices are subject to significant revision. Prior to implementing land disposal of waste residue (including waste sludge), consult with environmental regulatory agencies for guidance on acceptable disposal practices.
Homopolymers and copolymers of 2-Hydroxyethyl methacrylate is an indirect food additive for use only as a component of adhesives.
Combustible. Above 97 °C explosive vapour/air mixtures may be formed.
|Warning|H315: Causes skin irritation [Warning Skin corrosion/irritation]|P261, P264, P272, P280, P302+P352, P305+P351+P338, P321, P332+P313, P333+P313, P337+P313, P362, P363, and P501|H315 (91.26%): Causes skin irritation [Warning Skin corrosion/irritation]|Aggregated GHS information provided by 1832 companies from 12 notifications to the ECHA C&L Inventory. Each notification may be associated with multiple companies.|H317: May cause an allergic skin reaction [Warning Sensitization, Skin]|P261, P264, P272, P280, P302+P352, P305+P351+P338, P321, P333+P313, P337+P313, P363, and P501
Use water spray, dry powder, alcohol-resistant foam. In case of fire: keep drums, etc., cool by spraying with water.
SUITABLE PROTECTIVE CLOTHING & SELF-CONTAINED RESP PROTECTIVE APPARATUS SHOULD BE AVAILABLE FOR USE OF THOSE WHO MAY HAVE TO RESCUE PERSONS OVERCOME BY FUMES. /ACRYLIC ACID & DERIVATIVES/|HYDROXYETHYLMETHACRYLATE WAS DEMONSTRATED TO PASS THROUGH VINYL GLOVES.
30% GRADE (WITH XYLENE) IS FLAMMABLE; MODERATE FIRE RISK.
... Hazard is the generation of considerable exothermic heat in some of the reactions, so that high pressures & temp may develop. This danger ... should be borne in mind when designing plant. Awareness of the dangers and of good engineering design are essential to safety. Employees should be instructed about the necessity of cleansing the skin if it is contaminated by materials which are irritants or skin-absorbed. With careful design, however, and complete enclosure of those processes where toxic chemicals or intermediates occur, dangerous exposures can be avoided. /Acrylic acid & derivatives/|SRP: Contaminated protective clothing should be segregated in such a manner so that there is no direct personal contact by personnel who handle, dispose, or clean the clothing. Quality assurance to ascertain the completeness of the cleaning procedures should be implemented before the decontaminated protective clothing is returned for reuse by the workers. Contaminated clothing should not be taken home at end of shift, but should remain at employee's place of work for cleaning.|SRP: The scientific literature for the use of contact lenses in industry is conflicting. The benefit or detrimental effects of wearing contact lenses depend not only upon the substance, but also on factors including the form of the substance, characteristics and duration of the exposure, the uses of other eye protection equipment, and the hygiene of the lenses. However, there may be individual substances whose irritating or corrosive properties are such that the wearing of contact lenses would be harmful to the eye. In those specific cases, contact lenses should not be worn. In any event, the usual eye protection equipment should be worn even when contact lenses are in place.
Irritating to eyes and skin.
Personal protection: chemical protection suit and filter respirator for organic gases and vapours adapted to the airborne concentration of the substance. Collect leaking liquid in covered containers. Absorb remaining liquid in sand or inert absorbent. Then store and dispose of according to local regulations.
Store only if stabilized. Keep in the dark. Cool. Ventilation along the floor.
No indication can be given about the rate at which a harmful concentration of this substance in the air is reached.
The substance is irritating to the eyes, skin and respiratory tract. If this liquid is swallowed, aspiration into the lungs may result in chemical pneumonitis.
Repeated or prolonged contact may cause skin sensitization.
NO open flames. Above 97 °C use a closed system and ventilation.
PREVENT GENERATION OF MISTS!
Use ventilation (not if powder), local exhaust or breathing protection.
Protective gloves. Protective clothing.
Wear safety spectacles or eye protection in combination with breathing protection.
Toxicity
LD50 MICE INTRAPERITONEAL 0.497 ML/KG|LD50 MICE ORAL 5.1 ML/KG|LD50 Rat oral 11.2 g/kg|LD50 Rabbit percutaneous > 3.0 g/kg|For more Non-Human Toxicity Values (Complete) data for 2-HYDROXYETHYL METHACRYLATE (7 total), please visit the HSDB record page.
2-Hydroxyethyl methacrylate's production and use in acrylic resins, enamels, and as a binder for non-woven fabrics(1) may result in its release to the environment through various waste streams(SRC).
TERRESTRIAL FATE: Based on a classification scheme(1), an estimated Koc value of 43(SRC), determined from a log Kow of 0.47(2) and a regression-derived equation(3), indicates that 2-hydroxyethyl methacrylate is expected to have very high mobility in soil(SRC). Volatilization of 2-hydroxyethyl methacrylate from moist soil surfaces is not expected to be an important fate process(SRC) given an estimated Henry's Law constant of 4.6X10-9 atm-cu m/mole(SRC), using a fragment constant estimation method(4). 2-Hydroxyethyl methacrylate is not expected to volatilize from dry soil surfaces(SRC) based upon a vapor pressure of 0.126 mm Hg(5). Screening tests indicate that 2-hydroxyethyl methacrylate is readily biodegradable; it reached 92-100% of its theoretical BOD in 2 weeks using an activated sludge inoculum(6).|AQUATIC FATE: Based on a classification scheme(1), an estimated Koc value of 43(SRC), determined from a log Kow of 0.47(2) and a regression-derived equation(3), indicates that 2-hydroxyethyl methacrylate is not expected to adsorb to suspended solids and sediment in water(SRC). Volatilization from water surfaces is not expected(3) based upon an estimated Henry's Law constant of 4.6X10-9 atm-cu m/mole(SRC), developed using a fragment constant estimation method(4). According to a classification scheme(5), an estimated BCF of 1.3(SRC), from the log Kow(2), suggests the potential for bioconcentration in aquatic organisms is low. Hydrolysis of 2-hydroxyethyl methacrylate may be a significant process under basic conditions based upon a hydrolytic half-life of 4 hours at pH 11 for the structurally similar butyl acrylate(6); half-lives for butyl acrylate at pH 7, 8, and 9 were 4 years, 150 days, and 15 days(SRC), respectively, based upon a hydrolytic rate constant of 0.053 L/mole-sec(8). Screening tests indicate that 2-hydroxyethyl methacrylate is readily biodegradable; it reached 92-100% of its theoretical BOD in 2 weeks using an activated sludge inoculum(7).|ATMOSPHERIC FATE: According to a model of gas/particle partitioning of semivolatile organic compounds in the atmosphere(1), 2-hydroxyethyl methacrylate, which has a vapor pressure of 0.126 mm Hg at 25 °C(2), is expected to exist solely as a vapor in the ambient atmosphere. Vapor-phase 2-hydroxyethyl methacrylate is degraded in the atmosphere by reaction with photochemically-produced hydroxyl radicals(SRC); the half-life for this reaction in air is estimated to be 16 hours(SRC) from its estimated rate constant of 2.4X10-11 cu cm/molecule-sec at 25 °C(3). Because the structurally similar ethyl methacrylate does not absorb light in the environmental spectrum above 290 nm(4), direct photolysis of 2-hydroxyethyl methacrylate is not expected to occur(SRC).
The rate constant for the vapor-phase reaction of 2-hydroxyethyl methacrylate with photochemically-produced hydroxyl radicals has been estimated as 2.4X10-11 cu cm/molecule-sec at 25 °C(SRC) using a structure estimation method(1). This corresponds to an atmospheric half-life of about 16 hours at an atmospheric concentration of 5X10+5 hydroxyl radicals per cu cm(1). The rate constant for the vapor-phase reaction of 2-hydroxyethyl methacrylate with ozone has been estimated as 1.1X10-17 cu cm/molecule-sec at 25 °C(SRC) using a structure estimation method(1). This corresponds to an atmospheric half-life of about 1 day at an atmospheric concentration of 7X10+11 molecules/cu cm(1). Hydrolysis of 2-hydroxyethyl methacrylate may be a significant process under alkaline conditions based upon a hydrolytic half-life of 4 hours at pH 11 for the structurally similar butyl acrylate(2); half-lives for butyl acrylate at pH 7, 8, and 9 were 4 years, 150 days, and 15 days(SRC), respectively, based upon a hydrolytic rate constant of 0.053 L/mole-sec(4). 2-Hydroxyethyl methacrylate is not expected to directly photolyze in sunlight(SRC) based upon the lack of absorption of light at wavelengths >290 nm by the structurally similar ethyl methacrylate(3).
An estimated BCF of 1.3 was calculated for 2-hydroxyethyl methacrylate(SRC), using a log Kow of 0.47(1). According to a classification scheme(2), this BCF suggests the potential for bioconcentration in aquatic organisms is low.
The Koc of 2-hydroxyethyl methacrylate is estimated as approximately 43(SRC), using a log Kow of 0.47(1) and a regression-derived equation(2). According to a classification scheme(3), this estimated Koc value suggests that 2-hydroxyethyl methacrylate is expected to have very high mobility in soil.
The Henry's Law constant for 2-hydroxyethyl methacrylate is estimated as 4.6X10-9 atm-cu m/mole(SRC) using a fragment constant estimation method(1). This Henry's Law constant indicates that 2-hydroxyethyl methacrylate is expected to be essentially nonvolatile from water surfaces(2) and from moist soil surfaces(SRC). 2-Hydroxyethyl methacrylate is not expected to volatilize from dry soil surfaces(SRC) based upon a vapor pressure of 0.126 mm Hg(3).
NIOSH (NOES Survey 1981-1983) has statistically estimated that 16,032 workers (5,271 of these are female) are potentially exposed to 2-hydroxyethyl methacrylate in the US(1). Occupational exposure to 2-hydroxyethyl methacrylate may occur through inhalation and dermal contact with this compound at workplaces where 2-hydroxyethyl methacrylate is produced or used(SRC). The general population may be exposed to 2-hydroxyethyl methacrylate via inhalation or dermal contact with acrylic resins or enamels containing this compound(2).
Drug Information
SMALL QUANTITIES OF METHACRYLATES MAY READILY BE METABOLIZED BY SAPONIFICATION INTO THE ALCOHOL AND METHACRYLIC ACID. THE LATTER MAY FORM AN ACETYL-COENZYME DERIVATIVE, WHICH THEN ENTERS THE NORMAL LIPID METABOLISM. /METHACRYLATES/
Fresh air, rest. Refer for medical attention.
First rinse with plenty of water for at least 15 minutes, then remove contaminated clothes and rinse again. Refer for medical attention .
First rinse with plenty of water for several minutes (remove contact lenses if easily possible), then refer for medical attention.
Basic treatment: Establish a patent airway. Suction if necessary. Watch for signs of respiratory insufficiency and assist ventilations if necessary. Administer oxygen by nonrebreather mask at 10 to 15 L/min. Monitor for pulmonary edema and treat if necessary ... . Monitor for shock and treat if necessary ... . For eye contamination, flush eyes immediately with water. Irrigate each eye continuously with normal saline during transport ... . Do not use emetics. For ingestion, rinse mouth and administer 5 mL/kg up to 200 mL of water for dilution if the patient can swallow, has a strong gag reflex, and does not drool. Administer activated charcoal ... . /Esters and related compounds/|Advanced treatment: Consider orotracheal or nasotracheal intubation for airway control in the patient who is unconscious. Positive-pressure ventilation techniques with a bag-valve-mask device may be beneficial. Monitor cardiac rhythm and treat arrhythmias if necessary ... . Start an IV with D5W TKO /SRP: "To keep open", minimal flow rate/. Use lactated Ringer's if signs of hypovolemia are present. Watch for signs of fluid overload. For hypotension with signs of hypovolemia, administer fluid cautiously. Consider vasopressors if patient is hypotensive with a normal fluid volume. Watch for signs of fluid overload ... . Consider drug therapy for pulmonary edema ... . Use proparacaine hydrochloride to assist eye irrigation ... . /Esters and related compounds/
LAB TECHNICIAN DEVELOPED ALLERGIC CONTACT DERMATITIS ASSOC WITH NAUSEA, DIARRHEA, & PERSISTENT PARESTHESIA OF FINGERTIPS. HYDROXYETHYLMETHACRYLATE WAS DEMONSTRATED TO PASS THROUGH VINYL GLOVES.|2-Hydroxy-ethyl methacrylate and di- and tetra-ethylene glycol dimethacrylates have produced contact sensitization in workers in the printing industry.
2-hydroxyethyl methacrylate
Cough. Sore throat.
Redness.
Redness.
2-Hydroxyethyl methacrylate Use and Manufacturing
The methacrylates can be synthesized by catalytic oxidation of isobutylene and subsequent esterification with the appropriate alcohol, or by reacting acetone with hydrocyanic acid and subsequent esterification in sulfuric acid with the appropriate alcohol. /Methacrylic esters/|REACTION OF METHACRYLIC ACID WITH ETHYLENE OXIDE; TRANSESTERIFICATION OR ESTERIFICATION OF ETHYLENE GLYCOL WITH EITHER METHYL METHACRYLATE OR METHACRYLIC ACID.|Hydroxyalkyl methacrylates are prepared by the addition of methacrylic acid to the epoxide ring of alkylene oxides to form hydroxyalkyl methacrylates, ie, beta-hydroxyethyl and hydroxypropyl methacrylate. Oxides other than ethylene oxide give isomeric products; commercial hydroxypropyl methacrylate has about a 2-to-1 ratio of 2-hydroxypropyl methacrylate to 1-methyl-2-hydroxyethyl methacrylate. Effective catalysts include tertiary amines, anion-exchange resins, ferric chloride, and lithium methacrylate.
Acrylic resins, binder for nonwoven fabrics, enamels.
Adhesives and sealant chemicals
Adhesives and sealants
10,000,000 - 50,000,000 lb|(1982) 3.63-5.45X10+9 G (EST)
The 30% solution is made with xylene. Grades: 30%; 96%.|An inhibitor is usually added to solutions to prolong shelf life.|Commercial products contain hydroquinone methyl ether or similar polymerization inhibitors.
Adhesive manufacturing|2-Propenoic acid, 2-methyl-, 2-hydroxyethyl ester: ACTIVE|METHYL METHACRYLATE, AND IN GENERAL THE METHACRYLIC ESTERS, POLYMERIZE MUCH LESS READILY THAN THE CORRESPONDING ORDINARY ACRYLATES. NONE THE LESS, THEY ARE STABILIZED BY ADDING HYDROQUINONE OR PYROGALLOL, PARTICULARLY IN THE PRESENCE OF METALLIC COPPER. /METHACRYLATES/|In hydrogel contact lenses
Thin-layer chromatography (TLC), polarography, and spectrometry are used for soln measurements. Methacrylates in air have been analyzed by TLC, polarography, and colorimetry. Polarography has been used for determination of any residual monomer in the polymer. A variety of spectroscopic techniques, eg, NMR, IR, and Raman spectroscopy also have been used, particularly for analysis of surgical cements and dental restorative resins. /Methacrylic acid & derivatives/
Computed Properties
Molecular Weight:130.14
XLogP3:0.5
Hydrogen Bond Donor Count:1
Hydrogen Bond Acceptor Count:3
Rotatable Bond Count:4
Exact Mass:130.062994177
Monoisotopic Mass:130.062994177
Topological Polar Surface Area:46.5
Heavy Atom Count:9
Complexity:118
Covalently-Bonded Unit Count:1
Compound Is Canonicalized:Yes
Price Analysis
- Data: 2025-04-03
- Price: 15500.00Yuan/mt
- Change: 0
Recommended Suppliers of 2-Hydroxyethyl methacrylate
-
![China]() CN
CN
4 YRS
Business licensed Certified factoryManufactory Supplier of Dichloromethane,Linocaine hydrochloride -
![China]() CN
CN
4 YRS
Business licensed Certified factoryManufactory Supplier of fine chemicals,pharmaceutical materials -
![China]() CN
CN
9 YRS
Business licensedTrader Supplier of Phenoxyethanol -
![China]() CN
CN
4 YRS
Business licensed Certified factoryManufactory Supplier of HEA/HEMA/HPA/HPMAInquiryCAS No.: 868-77-9Grade: Industrial GradeContent: 98% -
![China]() CN
CN
2 YRS
Business licensedDistributor Supplier of HEMA,MAA
Latest News on 2-Hydroxyethyl methacrylate
- Specialty Chemicals Giant Synthomer Launches Innovation Center in Shanghai to Meet Chinese Market Needs
- Global consumption of specialty chemicals will add more than 100 billion U.S. dollars in five years
- U.S. Prince acquires specialty chemicals company Flow for US$2.1 billion
- Standard Industries acquires specialty chemicals company Grace for $7 billion
- Dow plans to establish a manufacturing base for specialty chemicals in South China
Learn More Other Chemicals
-
Ethylhexyl acrylate-methacrylic acid-methyl methacrylate copolymer
25133-98-6
-
acetic acid, [(4-methylphenyl)amino]oxo-, 2-hydroxyethyl e
1192806-63-5
-
2-Propenoic acid, 2-methyl-, 2-hydroxyethyl ester, polymer with butyl 2-propenoate, ethenylbenzene and methyl 2-methyl-2-propenoate
26588-80-7
-
2-Propenoic acid, 2-methyl-, 2-hydroxyethyl ester, polymer with ammonium 2-methyl-2-propenoate (1:1) Formula
68227-04-3
-
2-Propenoic acid, 2-methyl-, polymer with butyl 2-propenoate, 2-hydroxyethyl 2-propenoate and methyl 2-methyl-2-propenoate Formula
25230-94-8
-
2-Propenoic acid, 2-methyl-, 2-hydroxyethyl ester, polymer with butyl 2-propenoate and methyl 2-methyl-2-propenoate Formula
25951-39-7
-
2-Propenoic acid, 2-methyl-, butyl ester, polymer with ethenylbenzene, 2-hydroxyethyl 2-propenoate and methyl 2-methyl-2-propenoate Structure
63150-02-7
-
2-Propenoic acid, 2-methyl-, dodecyl ester, polymer with 2-hydroxyethyl 2-methyl-2-propenoate and methyl 2-methyl-2-propenoate Structure
29931-19-9
-
What is 2-Propenoic acid, 2-hydroxyethyl ester, reaction products with TDI
68479-07-2
-
What is 2-Propenoic acid, 2-methyl-, 2-hydroxyethyl ester, polymer with diethenylbenzene and methyl 2-methyl-2-propenoate
69631-31-8





