6-Bromo-2-methylquinoline
-
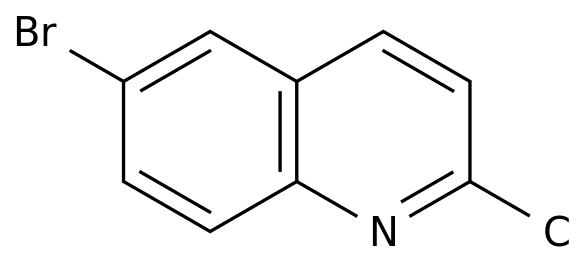
6-Bromo-2-methylquinoline
structure -
-
CAS No:
877-42-9
-
Formula:
C10H8BrN
-
Chemical Name:
6-Bromo-2-methylquinoline
-
Synonyms:
Quinoline,6-bromo-2-methyl-;Quinaldine,6-bromo-;6-Bromo-2-methylquinoline;6-Bromoquinaldine;2-Methyl-6-bromoquinoline
- Categories:
-
CAS No:
Characteristics
12.9
3.3
white to light yellow crystal powder
1.5±0.1 g/cm3
97 °C
299.7ºC at 760 mmHg
135.0±21.8 °C
1.654
Safety Information
NONH for all modes of transport
3
22-37/38-42-36/37/38-20/21/22
26-39-36/37/39-24/25-22
Xn,Xi
Irritant
P261-P280-P305 + P351 + P338-P342 + P311
H302-H315-H317-H318-H334-H335
|Danger|H302 (86.96%): Harmful if swallowed [Warning Acute toxicity, oral]|P261, P264, P270, P271, P272, P280, P285, P301+P312, P302+P352, P304+P340, P304+P341, P305+P351+P338, P310, P312, P321, P330, P332+P313, P333+P313, P337+P313, P342+P311, P362, P363, P403+P233, P405, and P501|Aggregated GHS information provided by 46 companies from 6 notifications to the ECHA C&L Inventory. Each notification may be associated with multiple companies.|H302 (100%): Harmful if swallowed [Warning Acute toxicity, oral]|P261, P264, P270, P271, P272, P280, P285, P301+P312, P302+P352, P304+P340, P304+P341, P305+P351+P338, P310, P312, P321, P330, P332+P313, P333+P313, P342+P311, P362, P363, P403+P233, P405, and P501|Aggregated GHS information provided by 11 companies from 1 notifications to the ECHA C&L Inventory. Each notification may be associated with multiple companies.
6-Bromo-2-methylquinoline Use and Manufacturing
General procedure: Amine (1.50 mmol) and aldehyde (1.00 mmol) were dissolved in 1.5 mL of Et2O in a reaction vial equipped with a magnetic stirrer bar, followed by the addition of Ag(I)-exchanged Montmorillonite K10 (0.50 g). After 5 min stirring, the solvent was removed in vacuo to obtain a dry powder. The reaction mixture was heated at a temperature of 120 oC for 3 h. After cooling to room temperature, the reaction mixture was filtered through a short silica plug and the solid residues washed well with CH2Cl2. The solvent was removed in vacuo and the crude product purified by column chromatography over silica gel eluting with a mixture of Hexane EtOAc to produce the title compounds (3a-3k). NOTE. For the recycle/reuse study, the crude product was gravity filtered and the clay material washed several times with CH2Cl2, air dried and weighed. This process was repeated for consecutive recycle/reuse reactions.A mixture of 4-bromobenzenamine (14.59g, 84.8mmol) and HCl (6N, 60ml) was heated to 100A mixture of 4-bromobenzenamine (14.59 g, 84.8 mmol) andHCl (6 N, 60 mL) was heated to 100 C, then crotonaldehyde (11.9 g, 170 mmol) was added slowly. The resulting mixture was refluxeduntil TLC shows no raw material exists. After cooling to roomtemperature, 200 mL H2O was added and the mixture wasextracted with EtOAc (100 mL 2) to remove the unreacted crotonaldehyde.The aqueous phase was neutralized with ammoniawater and then extracted with EtOAc (50 mL 2). The organicphase was dried over anhydrous Na2SO4 and evaporated to givecrude residue. The crude product was recrystallized in EtOAc/PE togive 13.46 g of 1 (60.64 mmol, 75.6percent).6-Bromo-2-methylquinoline. A mixture of 4-bromoaniline (1 g, 5.8 mmol), (2.pound.)-2-butenal (412 mg, 5.8 mmol), toluene (10 mL) and 6 N HCI (20 ml_) was refluxed overnight. The aqueous layer was separated, neutralized to PH = 7, extracted with ethyl acetate. The extract was dried over magnesium sulfate, concentrated in vacuo and purified via flash column chromatography (0-100percent ethyl acetate in hexanes) to give the title compound as a yellow solid (850 mg, 66percent). 1 H NMR (400 MHz, CHLOROFORM-d) δ ppm 8.00 (d, J=8.59 Hz, 1 H)7.92 - 7.98 (m, 2 H) 7.77 (dd, J=8.97, 2.15 Hz, 1 H) 7.34 (d, J=8.34 Hz, 1 H) 2.77 (s, 3 H).4-bromophenylamine (3.0 g, 17.44 mmol) was dissolved in 30 mL of 6N HCl. Crotonaldehyde (2.44 g, 34.88mmol) was slowly added thereto, and the reaction mixture was stirred at 100°C for 16 hours. The mixture was cooledto 0°C, neutralized with aqueous ammonia, extracted with EtOAc and purified by column chromatography to obtain thetitle compound (2 g, 52 percent).1H-NMR (CDCl3) δ 8.02∼7.92 (2H, m), 7.89 (1H, d), 7.73 (1H, d), 7.32 (1H, d), 1.60 (3H, s)To a solution of 4-bromoaniline (1.5 g, 8.72 mmol) in toluene (15 mL) was added cone. HC1 (15 mL). The solution was heated to 100 °C, then crotonaldehyde (1.44 mL, 17.44 mmol) was added dropwise and the mixture was stirred at 100° C for 3 h. The reaction mixture was then cooled to room temperature and basified with saturated aqueous NaOH until the pH was neutral. The aqueous layer was extracted with ethyl acetate (3 x 50 mL). The combined organic layers were washed with brine (50 mL), dried over Na1) Preparation of 6-bromo-2-methyl-quinolineDoebner-Miller synthesis:4-bromoaniline (12.4 g, 55.6 mmol) was added to a solution of 37percent HC1 at 0°C (24 mL). Paraldehyde (21 mL, 168 mmol, 3 eq) was then introduced and the mixture was left to react at room temperature for 1 hour, and then refluxed for 3 hours. After cooling to 0°C, sodium hydroxide (25 mL) was added dropwise, and the mixture was extracted with dichloromethane. The organic layer was washed twice with water and brine, then dried over MgS0p-Butyl bromide (10 g, 59 mmol) and vinyl ethyl ether (17 mL, 0·18 mol) were added to glacial acetic acid solvent (100 mL), and the mixture was stirred at 25 ° C for 3 to 4 hours. The reaction was detected by TLC. After the complete consumption of p-bromoaniline, the reaction was heated to reflux for 3 to 4 hours. After complete conversion of the reaction intermediate by TLC, the reaction system was poured into ice water (200 mL), and sodium hydrogencarbonate was adjusted to After extraction, the mixture was extracted with EtOAc (3×30 mL). The crude product was purified by silica gel column chromatography eluting with ΡΕ/ΕΑ = 8/1.A viscous liquid of 6.0 g was obtained in a yield of 46.1percent.Accurately weigh 0.005mmol (l. Lmg) of the transition metal, add 10mL has been put into the magnetic stirrer Young's reaction tube, The oxygen reaction was carried out in the Young's reaction tube, and the reaction was carried out under oxygen conditions. The syringe was applied to the Young's reaction tube0.04 mmol of co-catalyst I, 0.08 mmol of co-catalyst II, 0.2 mmol of 4-bromoaniline and 1 ml of absolute ethanol were added, The above-mentioned Young's reaction tube was placed on a magnetic stirrer and stirred at 150 ° C for 16 hours. After the reaction was completed, the pH of the reaction solution was adjusted, The reaction solution into the left after the treatment, 2_ methyl _6_ desert - Quilin pure product, the rate of 35percent.
Computed Properties
Molecular Weight:222.08
XLogP3:3.3
Hydrogen Bond Acceptor Count:1
Exact Mass:220.98401
Monoisotopic Mass:220.98401
Topological Polar Surface Area:12.9
Heavy Atom Count:12
Complexity:160
Covalently-Bonded Unit Count:1
Compound Is Canonicalized:Yes
Recommended Suppliers of 6-Bromo-2-methylquinoline
-
![China]() CN
CN
4 YRS
Business licensedTrader Supplier of Intermediates,Building blocks,API,Silicones,Peptides,Lab chemicals,Biochemicals,Pharmaceuticals,Screening Compounds,Food Additives -
![China]() CN
CN
3 YRS
Business licensedTrader Supplier of 1H,1H,2H,2H-PERFLUORODECYLTRIMETHOXYSILANEInquiryCAS No.: 877-42-9Grade: Industrial GradeContent: 99%
Learn More Other Chemicals
-
(2E)-3-(1-METHYL-1H-PYRROL-2-YL)ACRYLIC ACID
51485-76-8
-
Benadryl N-oxide hydrochloride
13168-00-8
-
6-CHLORO-3-IODO-IMIDAZO[1,2-A]PYRIDINE
885275-59-2
-
(2-Bromophenyl)diphenylphosphine Formula
62336-24-7
-
1-Morpholinocyclopentene Formula
936-52-7
-
4-[2-(Boc-amino)ethoxy]-benzoic acid Formula
168892-66-8
-
3-amino-5-bromopyridine-2-carboxylic acid Structure
870997-85-6
-
2-Amino-6-methylpyridine Structure
1824-81-3
-
What is 3-Bromo-2-methylthiophene
30319-05-2
-
What is THIOPHEN-2-YLMETHYL-PHOSPHONICACIDDIETHYLESTER
2026-42-8





