2-Methyl-1-butene
-
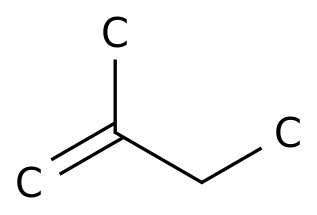
2-Methyl-1-butene
structure -
-
CAS No:
563-46-2
-
Formula:
C5H10
-
Chemical Name:
2-Methyl-1-butene
-
Synonyms:
1-Butene,2-methyl-;2-Methyl-1-butene;γ-Isoamylene;1-Isoamylene;NSC 72414;2-Methyl-1-butylene
- Categories:
-
CAS No:
Description
colourless liquid
2-methyl-1-butene appears as a colorless volatile liquid with a disagreeable odor. Insoluble in water and less dense than water. Vapors are heavier than air. Used to make other chemicals.|Liquid
2-methyl-1-butene appears as a colorless volatile liquid with a disagreeable odor. Insoluble in water and less dense than water. Vapors are heavier than air. Used to make other chemicals.|2-methylbut-1-ene is an alkene that is but-1-ene carrying a methyl substituent at position 2.
2-Methyl-1-butene Basic Attributes
70.13
70.13
209-250-7
33C9Y0I55H
72414
2459|2371
DTXSID1040249|DTXSID3052224
Colorless liquid
29012990
Characteristics
0
2.92
2-methyl-1-butene appears as a colorless volatile liquid with a disagreeable odor. Insoluble in water and less dense than water. Vapors are heavier than air. Used to make other chemicals.
0.6504 g/cm3 @ Temp: 20 °C
-137.5 °C
31.2 °C
31-32°C
1.384
soluble in ether, ethanol and benzene. Insoluble in water.
2-8°C
9.98 psi ( 20 °C)
Disagreeable odor
6.10e-11 cm3/molecule*sec
Hydroxyl radical rate constant= 6.07X10-11 cu cm/molecule-sec @ 25 °C
Highly flammable. Insoluble in water.
Hydrocarbons, Aliphatic Unsaturated
Highly Flammable
The unsaturated aliphatic hydrocarbons, such as 2-METHYL-1-BUTENE, are generally much more reactive than the alkanes. Strong oxidizers may react vigorously with them. Reducing agents can react exothermically to release gaseous hydrogen. In the presence of various catalysts (such as acids) or initiators, compounds in this class can undergo very exothermic addition polymerization reactions.
Safety Information
I
3
UN 2459 3/PG 1
3
12-65
16-62-33-29-7/9
F+,Xn,Xi
Irritant
Stable. Incompatible with oxidizing agents. Extremely flammable.
P210-P301 + P310-P331
H224-H304
SRP: At the time of review, criteria for land treatment or burial (sanitary landfill) disposal practices are subject to significant revision. Prior to implementing land disposal of waste residue (including waste sludge), consult with environmental regulatory agencies for guidance on acceptable disposal practices.
Excerpt from ERG Guide 128 [Flammable Liquids (Water-Immiscible)]: HIGHLY FLAMMABLE: Will be easily ignited by heat, sparks or flames. Vapors may form explosive mixtures with air. Vapors may travel to source of ignition and flash back. Most vapors are heavier than air. They will spread along ground and collect in low or confined areas (sewers, basements, tanks). Vapor explosion hazard indoors, outdoors or in sewers. Those substances designated with a (P) may polymerize explosively when heated or involved in a fire. Runoff to sewer may create fire or explosion hazard. Containers may explode when heated. Many liquids are lighter than water. Substance may be transported hot. For hybrid vehicles, ERG Guide 147 (lithium ion batteries) or ERG Guide 138 (sodium batteries) should also be consulted. If molten aluminum is involved, refer to ERG Guide 169. (ERG, 2016)|Flammable - 4th degree, Reactive - 1st degree
|Danger|H225 (96.3%): Highly Flammable liquid and vapor [Danger Flammable liquids]|P210, P233, P240, P241, P242, P243, P264, P270, P280, P301+P312, P303+P361+P353, P330, P370+P378, P403+P235, and P501|Aggregated GHS information provided by 27 companies from 3 notifications to the ECHA C&L Inventory. Each notification may be associated with multiple companies.|H224 (62.2%): Extremely flammable liquid and vapor [Danger Flammable liquids]|P201, P202, P210, P233, P240, P241, P242, P243, P261, P264, P270, P271, P273, P280, P281, P301+P310, P301+P312, P302+P352, P303+P361+P353, P304+P312, P304+P340, P305+P351+P338, P308+P313, P312, P321, P330, P331, P332+P313, P337+P313, P362, P370+P378, P391, P403+P233, P403+P235, P405, and P501|Aggregated GHS information provided by 127 companies from 6 notifications to the ECHA C&L Inventory. Each notification may be associated with multiple companies.
Excerpt from ERG Guide 128 [Flammable Liquids (Water-Immiscible)]: As an immediate precautionary measure, isolate spill or leak area for at least 50 meters (150 feet) in all directions. LARGE SPILL: Consider initial downwind evacuation for at least 300 meters (1000 feet). FIRE: If tank, rail car or tank truck is involved in a fire, ISOLATE for 800 meters (1/2 mile) in all directions; also, consider initial evacuation for 800 meters (1/2 mile) in all directions. (ERG, 2016)
Excerpt from ERG Guide 128 [Flammable Liquids (Water-Immiscible)]: ELIMINATE all ignition sources (no smoking, flares, sparks or flames in immediate area). All equipment used when handling the product must be grounded. Do not touch or walk through spilled material. Stop leak if you can do it without risk. Prevent entry into waterways, sewers, basements or confined areas. A vapor-suppressing foam may be used to reduce vapors. Absorb or cover with dry earth, sand or other non-combustible material and transfer to containers. Use clean, non-sparking tools to collect absorbed material. LARGE SPILL: Dike far ahead of liquid spill for later disposal. Water spray may reduce vapor, but may not prevent ignition in closed spaces. (ERG, 2016)
Excerpt from ERG Guide 128 [Flammable Liquids (Water-Immiscible)]: Wear positive pressure self-contained breathing apparatus (SCBA). Structural firefighters' protective clothing will only provide limited protection. (ERG, 2016)
HIGHLY FLAMMABLE, DANGEROUS FIRE ... RISK|Very dangerous fire hazard when exposed to heat, flame, or oxidizers.
To fight fire, use alcohol foam, mist, spray, dry chemical, carbon dioxide.
/GUIDE 128: FLAMMABLE LIQUIDS (NON-POLAR/WATER-IMMISCIBLE)/ Fire or Explosion: HIGHLY FLAMMABLE: Will be easily ignited by heat, sparks or flames. Vapors may form explosive mixtures with air. Vapors may travel to source of ignition and flash back. Most vapors are heavier than air. They will spread along ground and collect in low or confined areas (sewers, basements, tanks). Vapor explosion hazard indoors, outdoors or in sewers. Those substances designated with a "P" may polymerize explosively when heated or involved in a fire. Runoff to sewer may create fire or explosion hazard. Containers may explode when heated. Many liquids are lighter than water. Substances may be transported hot.|/GUIDE 128: FLAMMABLE LIQUIDS (NON-POLAR/WATER-IMMISCIBLE)/ Health: Inhalation or contact with material may irritate or burn skin and eyes. Fire may produce irritating, corrosive and/or toxic gases. Vapors may cause dizziness or suffocation. Runoff from fire control or dilution water may cause pollution.|/GUIDE 128: FLAMMABLE LIQUIDS (NON-POLAR/WATER-IMMISCIBLE)/ Public Safety: CALL Emergency Response Telephone Number ... . As an immediate precautionary measure, isolate spill or leak area for at least 50 meters (150 feet) in all directions. Keep unauthorized personnel away. Stay upwind. Keep out of low areas. Ventilate closed spaces before entering.|/GUIDE 128: FLAMMABLE LIQUIDS (NON-POLAR/WATER-IMMISCIBLE)/ Protective Clothing: Wear positive pressure self-contained breathing apparatus (SCBA). Structural firefighters' protective clothing will only provide limited protection.|For more DOT Emergency Guidelines (Complete) data for 2-METHYL-1-BUTENE (8 total), please visit the HSDB record page.
No person may /transport,/ offer or accept a hazardous material for transportation in commerce unless that person is registered in conformance ... and the hazardous material is properly classed, described, packaged, marked, labeled, and in condition for shipment as required or authorized by ... /the hazardous materials regulations (49 CFR 171-177)./|The International Maritime Dangerous Goods Code lays down basic principles for transporting hazardous chemicals. Detailed recommendations for individual substances and a number of recommendations for good practice are included in the classes dealing with such substances. A general index of technical names has also been compiled. This index should always be consulted when attempting to locate the appropriate procedures to be used when shipping any substance or article.
| 2 - Materials that, under emergency conditions, can cause temporary incapacitation or residual injury.| 4 - Materials that rapidly or completely vaporize at atmospheric pressure and normal ambient temperature or that are readily dispersed in air and burn readily.| 0 - Materials that in themselves are normally stable, even under fire conditions.
Exhaust from turbojet engines operated at simulated high-altitude flight conditions contained 0.10-5.35 ppmC (ppm carbon) of 2-methyl-1-butene(1). Exhaust from a two-stroke moped contained 0.16 and 0.35 % weight of 2-methyl-1-butene, using an alkalate-based fuel(2). 2-Methyl-1-butene was emitted in the exhaust of 10 four-stroke lawnmower engines at 0.28 and 0.29 % of the total organic emissions, for two different gasolines(3). 2-Methyl-1-butene was emitted in the exhaust of a single-cylinder, production-type engine run on gasoline at 8 ppmC(4).|2-Methyl-1-butene accounted for 0.12 % of the total United Kingdom volatile organic compound emissions (by mass distribution) in 1990(1). Emissions of 2-methyl-1-butene from motor vehicles, measured inside the Van Nuys Tunnel, Los Angeles, CA in 1993 were determined to average 35 mg/L fuel consumed (2). 2-Methyl-1-butene was measured in the exhaust of 67 vehicles representative of the Sydney, Australia fleet at an average concentration of 0.3 %wt/wt non-methane hydrocarbons (3). 2-Methyl-1-butene was detected in the Tingstad Tunnel, Goteborg, Sweden at 0.17 to 0.33 % of the total non-methane volatile hydrocarbons over 5 different sampling days during 1991 and 1992 (4). Light-duty vehicle emissions measured at the Caldecott Tunnel, San Francisco, CA during August and October, 1994, contained 2-methyl-1-butene at 0.23 and 0.34 weight % of total volatile organic compounds; August fuel is low oxygenate concentration, October fuel is high oxygenate concentration(5).
SOURCE DOMINATED: Trace amounts of 2-methyl-1-butene were detected in brush fire smoke Cajon Pass, CA. 2-Methyl-1-butene was also detected in samples from an industrial area of Los Angeles County, CA, a Riverside, CA shopping center parking lot, and a nearby busy intersection with three filling stations. The compound was not detected in smog taken from the University of California Riverside campus(1). Air samples taken downwind from a natural gas facility at Piceance Creek in Rio Blanco County, CO, contained 2-methyl-1-butene at 10.7 ppbC(2). Monitoring data was collected from 6 self-service stations in the US (Cincinnati, OH, Phenoix, AZ, Los Angeles, CA) to assess consumer exposure to airborne gasoline components(3). 2-Methyl-1-butene was detected (detection of quantitation = 3 ug/sample) at a range of 0.02-5.2 mg/cu m(3).|URBAN/SUBURBAN: Weekday air samples were taken from 6 to 9 am in 39 US cities during a 1984 through 1986 time period(1). In 822 samples, 2-methyl-1-butene was detected at a median concn of 2.6 ppbC, with a maximum value of 242 ppbC(1). Ambient air from Tulsa, OK contained 2-methyl-1-butene at a range of 0.6-6.4 ppbC(2). During a severe Los Angeles area photochemical smog episode in September 1993, 2-methyl-1-butene was measured in the vapor phase at an average concentration of 1.13 ug/cu m (range not detected to 3.40 ug/cu m)(3). Air samples collected from the industrial area of Houston, TX, contained 2-methyl-1-butene at concentrations of not detected to 53.8, 8.7 to 47.6 and not detected to 3.7 ppbC on separate days in September 1973, January 1974, and April, 1974, respectively (detection limit not reported) (4). Air samples collected by aircraft over Miami in May 1976 contained 2-methyl-1-butene at concentrations of 0.5 and 0.6 ppbC(5). Air samples collected from downtown Los Angeles during morning rush hour, fall 1968, contained 2-methyl-1-butene at 15 ppbC(6). The average concentration of 2-methyl-1-butene in downtown Porto Alegre, Brazil over the period March 1996 to April 1997 is 4.4 mg/cu m (n=46)(7). Concentrations of 2-methyl-1-butene inside and outside the town of Boone, NC (university town, little industry) from September 1981 to October 1982 ranged from 1.5 to 3.7 and 0.3 to 0.5 ppbC, respectively(8). The average concentration of 2-methyl-1-butene is 0.5 ppbv (0.5 weight %; n=140) in the morning air of Sydney, 30 day monitoring period at 3 different sampling sites (1 downwind of central business district, 2 sites downwind of a refinery-petrochemical complex)(9). 2-Methyl-1-butene was present at 2.0 ppb in ambient air samples collected at the Riverside CA County Building of Health and Finance between August 1965 and March 1966 in the early morning(10). The mean concentration of 2-methyl-1-butene measured between 6 and 9 am during the summers of 1984 to 1986 in 39 US cities located between 26 deg N and 43 deg N latitude was 2.6 ppbC(11).|RURAL/REMOTE: 2-Methyl-1-butene was detected in the ambient air of Sevier County, TN in the Great Smokey Mountains National Park, ranging from not detected to 1.1 ppbC(1). Air samples collected in the remote Piceance Creek Basin, Colorado contained 2-methyl-1-butene at concentrations from 0.4 to 1.2 ppbC(1). The concentration of 2-methyl-1-butene in air samples collected above a mixed deciduous forest in southern Ontario, Canada in 1993 was 0.002 and 0.003 ppbv in day and nighttime samples, respectively(2). 2-Methyl-1-butene was measured at 0.3 and 0.4 ppbC in ground level air samples collected in an orange grove during May near Dunedin, FL(3). An average 2-methyl-1-butene concentration of 0.2 mg/cu m was measured 30 km southeast of Porto Alegre, Brazil (upwind of the city during sample collection) in November 1996(4). Concentrations of 2-methyl-1-butene at 5 remote locations in the southern Appalachian Mountains of northwestern North Carolina from September 1981 to October 1982 ranged from not detected to 0.4 ppbC(5). 2-Methyl-1-butene was measured in air samples at concentrations from 0.2 to 3.2 ppbC collected from a picnic area at Jones State Forest between January 4 to 6, 1978(6).
Toxicity
2-Methyl-1-butene's production and use in organic synthesis and pesticide formulations(1) result in its release to the environment through various waste streams(SRC). 2-Methyl-1-butene has been identified in emissions from automobiles, turbines, gasoline, and forest fires(2).
TERRESTRIAL FATE: Based on a classification scheme(1), an estimated Koc value of 68(SRC), determined from a structure estimation method(2), indicates that 2-methyl-1-butene should have high mobility in soil(SRC). Volatilization of 2-methyl-1-butene from moist soil surfaces is expected to be an important fate process(SRC) given an estimated Henry's Law constant of 0.43 atm-cu m/mole(SRC) calculated from its water solubility(4) and vapor pressure(3). The potential for volatilization of 2-methyl-1-butene from dry soil surfaces may exist(SRC) based upon a vapor pressure of 610 mm Hg(3). While biodegradation data in soil were not located, 2-methyl-1-butene's relatively low molecular weight, hydrocarbon structure suggests that biodegradation is expected to be an important fate process(5).|AQUATIC FATE: Based on a classification scheme(1), an estimated Koc value of 68(SRC), determined from an estimation method(2), indicates that 2-methyl-1-butene is not expected to adsorb to suspended solids and sediment in water(SRC). Volatilization from water surfaces is expected(3) based upon a Henry's Law constant of 0.43 atm-cu m/mole(SRC) calculated from its water solubility(5) and vapor pressure(4). Using this Henry's Law constant and an estimation method(3), volatilization half-lives for a model river and model lake are 2.4 hours and 3.3 days, respectively(SRC). According to a classification scheme(6), a BCF of 25(SRC) estimated from its estimated log Kow of 2.72(8) and a regression-derived equation(7), suggests the potential for bioconcentration in aquatic organisms is low. While biodegradation data in water were not located, 2-methyl-1-butene's relatively low molecular weight, hydrocarbon structure suggests that biodegradation is expected to be an important fate process(9).|ATMOSPHERIC FATE: 2-Methyl-1-butene which has a vapor pressure of 610 mm Hg at 25 °C(2), is a volatile organic compound and should exist solely as a vapor in the atmosphere(1). It is degraded in the atmosphere by reaction with photochemically-produced hydroxyl radicals and ozone molecules(SRC); the half-life for the reaction with hydroxyl radicals in air is estimated to be 6.3 hours(SRC), calculated from its rate constant of 6.07X10-11 cu cm/molecule-sec at 25 °C(3). The reaction of 2-methyl-1-butene with ozone is estimated to be much slower than that with hydroxyl radicals and therefore would not markedly alter 2-methyl-1-butene's atmospheric half-life.
The rate constant for the vapor-phase reaction of 2-methyl-1-butene with photochemically-produced hydroxyl radicals is 6.07X10-11 cu cm/molecule-sec at 25 °C(1). This corresponds to an atmospheric half-life of about 6.3 hours at an atmospheric concentration of 5X10+5 hydroxyl radicals per cu cm(2). The rate constant for the vapor-phase reaction of 2-methyl-1-butene with ozone in the atmosphere has been estimated as 1.2X10-17 cu cm/molecule-sec at 25 °C(SRC) using a structure estimation method(2). This corresponds to an atmospheric half-life of about 23 hours at an atmospheric concentration of 7X10+11 ozone molecules per cu cm(2). 2-Methyl-1-butene is not expected to undergo hydrolysis in the environment due to the lack of hydrolyzable functional groups(3) nor to directly photolyze due to the lack of absorption in the environmental UV spectrum (>290 nm)(3).|Olefins are known to contribute to photochemical smog; their atmospheric reactions contribute to the formation of ozone and other photochemical oxidants and free radicals. Photochemical ozone creation potentials from 57.7 to 96.3, relative to ethylene (=100), were calculated for 2-methyl-1-butene over a 5-day period from the peak ozone concentration reached in the afternoon for emissions in the United Kingdom(1). Products of the gas-phase reaction of ozone with 2-methyl-1-butene, in the presence of an hydroxyl radical scavenger (cyclohexane) were (product, yield): formaldehyde, 11.1; acetaldehyde, 14.7; acetone, 1.1; 2-butanone, 22.0; biacetyl, 19.0; cyclohexanone, 14.6; formic acid, 16.6(2). The gas-phase reaction of ozone with 2-methyl-1-butene in the absence and presence of oxygen produced 0.105 and 0.205 moles acetaldehyde formed per mole of ozone consumed, respectively, and 0.070 and 0.050 moles acetone formed per mole of ozone consumed, respectively, and 0.26 and 0.44 moles methyl ethyl ketone formed per mole of ozone consumed, respectively; no analysis for formaldehyde was conducted(3). The relative reaction rate of nitric oxide photooxidation in the presence of 2-methyl-1-butene, relative to the isobutene rate, is 0.97; the relative reaction rate of ozone in the presence of 2-methyl-1-butene, relative to the isobutene rate, is 1.3(4). Ambient air collected from the atmosphere in downtown Los Angeles, CA during the morning traffic peak hours was exposed to solar radiation; 90% conversion of 2-methyl-1-butene was reported in 4 hours (5). The photooxidation of acetaldehyde with 2-methyl-1-butene in air using sunlight reported in a 4 and 12% loss per hour of 2-methyl-1-butene and acetaldehyde, respectively; the photooxidation of the aldehyde is believed to generate reactive species which can then react with the hydrocarbon(6).
An estimated BCF of 25 was calculated for 2-methyl-1-butene(SRC), using an estimated log Kow of 2.72(1) and a regression-derived equation(2). According to a classification scheme(3), this BCF suggests the potential for bioconcentration in aquatic organisms is low.
Using a structure estimation method based on molecular connectivity indices(1), the Koc for 2-methyl-1-butene can be estimated to be 68(SRC). According to a classification scheme(2), this estimated Koc value suggests that 2-methyl-1-butene is expected to have high mobility in soil.
The Henry's Law constant for 2-methyl-1-butene calculated from its vapor pressure, 610 mm Hg(1), and water solubility, 130 mg/l(2), is 0.43 atm-cu m/mole (SRC). This Henry's Law constant indicates that 2-methyl-1-butene should volatilize rapidly from water surfaces(3). Based on this Henry's Law constant, the volatilization half-life from a model river (1 m deep, flowing 1 m/sec, wind velocity of 3 m/sec)(3) is estimated as 2.4 hours(SRC). The volatilization half-life from a model lake (1 m deep, flowing 0.05 m/sec, wind velocity of 0.5 m/sec)(3) is estimated as 3.3 days(SRC). 2-Methyl-1-butene's Henry's Law constant(1,2) indicates that volatilization from moist soil surfaces may occur(SRC). The potential for volatilization of 2-methyl-1-butene from dry soil surfaces exists based upon its vapor pressure of 610 mm Hg(1).
Occupational exposure to 2-methyl-1-butene may occur through inhalation and dermal contact with this compound at workplaces where 2-methyl-1-butene or fuels containing 2-methyl-1-butene is produced or used. The general population may be exposed to 2-methyl-1-butene via inhalation of gasoline or diesel fuel particularly during pumping of gasoline at self-serve stations. (SRC)
Drug Information
Excerpt from ERG Guide 128 [Flammable Liquids (Water-Immiscible)]: Inhalation or contact with material may irritate or burn skin and eyes. Fire may produce irritating, corrosive and/or toxic gases. Vapors may cause dizziness or suffocation. Runoff from fire control or dilution water may cause pollution. (ERG, 2016)
Excerpt from ERG Guide 128 [Flammable Liquids (Water-Immiscible)]: Ensure that medical personnel are aware of the material(s) involved and take precautions to protect themselves. Move victim to fresh air. Call 911 or emergency medical service. Give artificial respiration if victim is not breathing. Administer oxygen if breathing is difficult. Remove and isolate contaminated clothing and shoes. In case of contact with substance, immediately flush skin or eyes with running water for at least 20 minutes. Wash skin with soap and water. In case of burns, immediately cool affected skin for as long as possible with cold water. Do not remove clothing if adhering to skin. Keep victim calm and warm. (ERG, 2016)
Basic Treatment: Establish a patent airway. Suction if necessary. Watch for signs of respiratory insufficiency and assist ventilations if necessary. Administer oxygen by nonrebreather mask at 10 to 15 L/min. Monitor for pulmonary edema and treat if necessary ... Anticipate seizures and treat as necessary ... For eye contamination, flush eyes immediately with water. Irrigate each eye continuously with normal saline during transport ... Do not use emetics. For ingestion, rinse mouth and administer 5 mL/kg up to 200 mL of water for dilution if the patient can swallow, has a strong gag reflex, and does not drool. Administer activated charcoal ... Treat frostbite with rapid rewarming techniques ... /Aliphatic hydrocarbons and related compounds/|Advanced treatment: Consider orotracheal or nasotracheal intubation for airway control in the patient who is unconscious or in respiratory arrest. Positive-pressure ventilation techniques with a bag-valve-mask device may be beneficial. Monitor cardiac rhythm and treat arrhythmias as necessary ... Start an IV with D5W TKO /SRP: "To keep open", minimal flow rate/. Use lactated Ringer's if signs of hypovolemia are present. Watch for signs of fluid overload. Consider drug therapy for pulmonary edema ... Treat seizures with diazepam (Valium) ... Use proparacaine hydrochloride to assist eye irrigation ... /Aliphatic hydrocarbons and related compounds/
2-Methyl-1-butene Use and Manufacturing
Refinery gas|CATALYTIC CRACKING OR STEAM CRACKING OF PETROLEUM FOLLOWED BY ISOLATION OF C5 FRACTION AND EXTRACTION WITH COLD AQUEOUS SULFURIC ACID
Organic synthesis, pesticide formulations.
Fuels and fuel additives|Intermediates
Fuels and related products
50,000,000 - 100,000,000 lb|1,000,000 - 10,000,000 lb|(1977) AT LEAST 4.54X10+9 GRAMS|(1981) 7.43X10+10 G (TOTAL, MIXED PENTENES)
Grades: 95%, 99%, and research
Petroleum refineries|1-Butene, 2-methyl-: ACTIVE|Petrochemical manufacturing|Butene, 2-methyl-: ACTIVE
Fire Hazards -> Flammable - 4th degree, Reactive - 1st degree|Cosmetics -> Solvent
Computed Properties
Molecular Weight:70.13
XLogP3:2.5
Rotatable Bond Count:1
Exact Mass:70.078250319
Monoisotopic Mass:70.078250319
Heavy Atom Count:5
Complexity:35.3
Covalently-Bonded Unit Count:1
Compound Is Canonicalized:Yes
Learn More Other Chemicals
-
6-Methyl-2′,3′-dideoxyadenosine
85326-07-4
-
2-[[2-(1H-indol-3-yl)acetyl]-methylamino]-2-phenyl-N-(4-propan-2-ylphenyl)acetamide
853138-65-5
-
Phenylmethyl docosanoate
85263-74-7
-
5,6,7,8-Tetrahydro-2-naphthalenesulfonic acid Formula
93-12-9
-
6-CHLORO-3-PHENYL-2-QUINOLINOL Formula
85274-64-2
-
2,5-Furandione, dihydro-, monopolyisobutylene derivs. Formula
67762-77-0
-
N-(4,4,5,5,6,6,7,7,8,8,9,9,10,10,11,11,11-Heptadecafluoroundecyl)maleimide Structure
852527-40-3
-
2-Benzothiazolecarboxaldehyde,5-methoxy-(5CI) Structure
854059-90-8
-
What is Benzamide, 5-(aminosulfonyl)-N-[(2-ethyloctahydro-1H-isoindol-1-yl)methyl]-2-methoxy-, hydrochloride (1:1)
85409-39-8
-
What is 3-(2,4-Dimethylphenyl)-5-phenyl-1H-1,2,4-triazole
85303-90-8




