1,2-Difluoroethane
-
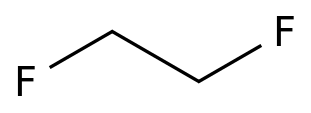
1,2-Difluoroethane
structure -
-
CAS No:
624-72-6
-
Formula:
C2H4F2
-
Chemical Name:
1,2-Difluoroethane
-
Synonyms:
Ethane,1,2-difluoro-;Freon 152;1,2-Difluoroethane;HFC 152;R 152 (refrigerant);R 152;HCFC 152
- Categories:
-
CAS No:
Characteristics
0
1.21 (est)
1.024 g/cm3 @ Temp: 10 °C
-118.59°C (estimate)
30.7 °C
1.254
In water, 2.31X10+3 mg/L at 25 deg C (est)
616 mm Hg at 25 deg C /from experimentally-derived coefficients/
Henry's Law constant = 3.87X10-1 atm-cu m/mol at 25 °C (est)
1.12e-13 cm3/molecule*sec
Dipole moment = 2.67 debye|Hydroxy radical reaction rate constant = 1.12X10-13 cu cm/molec-sec at 2t °C
Critical temperature = 107.5 °C
Safety Information
GAS, TOXIC, FLAMMABLE
3161
12
3/7-9-16-33
F,T
Highly Toxic/Flammable
P210, P233, P240, P241, P242, P243, P261, P271, P280, P303+P361+P353, P304+P312, P304+P340, P312, P370+P378, P403+P235, P501
H224
SRP: The most favorable course of action is to use an alternative chemical product with less inherent propensity for occupational harm/injury/toxicity or environmental contamination. Recycle any unused portion of the material for its approved use or return it to the manufacturer or supplier. Ultimate disposal of the chemical must consider: the material's impact on air quality; potential migration in soil or water; effects on animal and plant life; and conformance with environmental and public health regulations.|Because of recent discovery of potential ozone decomposition in the stratosphere by fluorotrichloromethane, this material should be released to the environment only as a last resort. Waste material should be /recovered and/ returned to the vendor, or to licensed waste disposal company.
|Danger|H224 (100%): Extremely flammable liquid and vapor [Danger Flammable liquids]|P210, P233, P240, P241, P242, P243, P261, P271, P280, P303+P361+P353, P304+P312, P304+P340, P312, P370+P378, P403+P235, and P501|Aggregated GHS information provided by 2 companies from 2 notifications to the ECHA C&L Inventory. Each notification may be associated with multiple companies.
Personnel protection: Wear appropriate chemical protective gloves and goggles. /Difluoroethane/|Neoprene gloves, protective clothing, and eye protection minimize risk of topical contact. Degreasing effect on skin can be treated with lanolin ointment. /Fluorocarbons/|Forced air ventilation at level of vapor concn together with use of individual breathing devices with independent air supply will minimize risk of inhalation. Lifelines should be worn when entering tanks or other confined spaces. /Fluorocarbons/
If material on fire or involved in fire: Do not extinguish fire unless flow can be stopped. Use water in flooding quantities as fog. Cool all affected containers with flooding quantities of water. Apply water from as far a distance as possible. /Difluoroethane/|Wear positive pressure self-contained breathing apparatus when fighting fires involving this material. Approach fire with caution. If fire becomes uncontrollable or container is exposed to direct flame consider evacuation of one-half (1/2) mile radius. /Difluoroethane/
SRP: Local exhaust ventilation should be applied wherever there is an incidence of point source emissions or dispersion of regulated contaminants in the work area. Ventilation control of the contaminant as close to its point of generation is both the most economical and safest method to minimize personnel exposure to airborne contaminants. Ensure that the local ventilation moves the contaminant away from the worker.|If material not on fire and not involved in fire: Keep sparks, flames, and other sources of ignition away. Keep material out of water sources and sewers. Attempt to stop leak if without undue personnel hazard. Use water spray to knock-down vapors. /Difluoroethane/|Personel protection: Avoid breathing vapors. Keep upwind. Do not handle broken packages unless wearing appropriate personal protective equipment. If material leaking (not on fire) consider evacuation from downwind area based on amount of material spilled, location and weather conditions. /Difluoroethane/|Sufficient exhaust and general ventilation should be provided to keep vapor concn below recommended levels. /Fluorocarbons/|For more Preventive Measures (Complete) data for 1,2-Difluoroethane (9 total), please visit the HSDB record page.
/GUIDE 115: GASES - FLAMMABLE (INCLUDING REFRIGERATED LIQUIDS)/ Fire or Explosion: EXTREMELY FLAMMABLE. Will be easily ignited by heat, sparks or flames. Will form explosive mixtures with air. Vapors from liquefied gas are initially heavier than air and spread along ground. ... Vapors may travel to source of ignition and flash back. Cylinders exposed to fire may vent and release flammable gas through pressure relief devices. Containers may explode when heated. Ruptured cylinders may rocket. /Difluoroethane/|/GUIDE 115: GASES - FLAMMABLE (INCLUDING REFRIGERATED LIQUIDS)/ Health: Vapors may cause dizziness or asphyxiation without warning. Some may be irritating if inhaled at high concentrations. Contact with gas or liquefied gas may cause burns, severe injury and/or frostbite. Fire may produce irritating and/or toxic gases. /Difluoroethane/|/GUIDE 115: GASES - FLAMMABLE (INCLUDING REFRIGERATED LIQUIDS)/ Public Safety: CALL Emergency Response Telephone Number... As an immediate precautionary measure, isolate spill or leak area for at least 100 meters (330 feet ) in all directions. Keep unauthorized personnel away. Stay upwind. Many gases are heavier than air and will spread along ground and collect in low or confined areas (sewers, basements, tanks). Keep out of low areas. /Difluoroethane/|/GUIDE 115: GASES - FLAMMABLE (INCLUDING REFRIGERATED LIQUIDS)/ Protective Clothing: Wear positive pressure self-contained breathing apparatus (SCBA). Structural firefighters' protective clothing will only provide limited protection. Always wear thermal protective clothing when handling refrigerated/cryogenic liquids. /Difluoroethane/|For more DOT Emergency Guidelines (Complete) data for 1,2-Difluoroethane (8 total), please visit the HSDB record page.
No person may /transport,/ offer or accept a hazardous material for transportation in commerce unless that person is registered in conformance ... and the hazardous material is properly classed, described, packaged, marked, labeled, and in condition for shipment as required or authorized by ... /the hazardous materials regulations (49 CFR 171-177)./|The International Maritime Dangerous Goods Code lays down basic principles for transporting hazardous chemicals. Detailed recommendations for individual substances and a number of recommendations for good practice are included in the classes dealing with such substances. A general index of technical names has also been compiled. This index should always be consulted when attempting to locate the appropriate procedures to be used when shipping any substance or article.
Toxicity
If inhalation occurs, epinephrine or other sympathomimetic amines and adrenergic activators should not be admin since they will further sensitize heart to development of arrhythmias. /Fluorocarbons/
It is possible that patients with cardiac or resp disorders may prove esp susceptible. /Fluorocarbons/
1,2-Difluoroethane's former production and use as a refrigerant(1) may have resulted in its release to the environment through various waste streams(SRC).
TERRESTRIAL FATE: Based on a classification scheme(1), an estimated Koc value of 40(SRC), determined from a structure estimation method(2), indicates that 1,2-difluoroethane is expected to have very high mobility in soil(SRC). Volatilization of 1,2-difluoroethane from moist soil surfaces is expected to be an important fate process(SRC) given an estimated Henry's Law constant of 0.39 atm-cu m/mole(SRC), using a fragment constant estimation method(3). 1,2-Difluoroethane is expected to volatilize from dry soil surfaces(SRC) based upon a vapor pressure of 616 mm Hg(4). Highly fluorinated compounds such as 1,2-difluoroethane are not expected to biodegrade rapidly(5).|AQUATIC FATE: Based on a classification scheme(1), an estimated Koc value of 40(SRC), determined from a structure estimation method(2), indicates that 1,2-difluoroethane is not expected to adsorb to suspended solids and sediment(SRC). Volatilization from water surfaces is expected(3) based upon an estimated Henry's Law constant of 0.39 atm-cu m/mole(SRC), developed using a fragment constant estimation method(4). Using this Henry's Law constant and an estimation method(3), volatilization half-lives for a model river and model lake are 2.4 hours and 3.2 days, respectively(SRC). According to a classification scheme(5), an estimated BCF of 3(SRC), from an estimated log Kow of 1.2(6) and a regression-derived equation(7), suggests the potential for bioconcentration in aquatic organisms is low(SRC). Hydrolysis is not expected to be an important process(SRC) based on estimated hydrolysis half-lives of 6.2X10+8 and 6.2X10+7 years at pHs 7 and 8, respectively(8). Highly fluorinated compounds such as 1,2-difluoroethane are not expected to biodegrade rapidly(9).|ATMOSPHERIC FATE: According to a model of gas/particle partitioning of semivolatile organic compounds in the atmosphere(1), 1,2-difluoroethane, which has a vapor pressure of 616 mm Hg at 25 °C(2), is expected to exist solely as a vapor in the ambient atmosphere. Vapor-phase 1,2-difluoroethane is degraded in the atmosphere by reaction with photochemically-produced hydroxyl radicals(SRC); the half-life for this reaction in air is estimated to be 140 to 180 days(SRC), calculated from its rate constants of 1.12X10-13(3) to 9.1X10-14(4) cu cm/molecule-sec. 1,2-Difluoroethane does not contain chromophores that absorb at wavelengths >290 nm(5) and therefore is not expected to be susceptible to direct photolysis by sunlight(SRC).
The rate constant for the vapor-phase reaction of 1,2-difluoroethane with photochemically-produced hydroxyl radicals is 1.12X10-13(1) to 9.1X10-14(2) cu cm/molecule-sec. This corresponds to an atmospheric half-life of about 140 to 180 days at an atmospheric concentration of 5X10+5 hydroxyl radicals per cu cm(1). A base-catalyzed second-order hydrolysis rate constant of 3.6X10-10 L/mole-sec(SRC) was estimated using a structure estimation method(3); this corresponds to half-lives of 6.2X10+8 and 6.2X10+7 years at pH values of 7 and 8, respectively(3). 1,2-Difluoroethane does not contain chromophores that absorb at wavelengths >290 nm(4) and therefore is not expected to be susceptible to direct photolysis by sunlight(SRC).
An estimated BCF of 3 was calculated in fish for 1,2-difluoroethane(SRC), using an estimated log Kow of 1.2(1) and a regression-derived equation(2). According to a classification scheme(3), this BCF suggests the potential for bioconcentration in aquatic organisms is low(SRC).
Using a structure estimation method based on molecular connectivity indices(1), the Koc of 1,2-difluoroethane can be estimated to be 40(SRC). According to a classification scheme(2), this estimated Koc value suggests that 1,2-difluoroethane is expected to have very high mobility in soil.
The Henry's Law constant for 1,2-difluoroethane is estimated as 0.39 atm-cu m/mole(SRC) using a fragment constant estimation method(1). This Henry's Law constant indicates that 1,2-difluoroethane is expected to volatilize rapidly from water surfaces(2). Based on this Henry's Law constant, the volatilization half-life from a model river (1 m deep, flowing 1 m/sec, wind velocity of 3 m/sec)(2) is estimated as 2.4 hours(SRC). The volatilization half-life from a model lake (1 m deep, flowing 0.05 m/sec, wind velocity of 0.5 m/sec)(2) is estimated as 3.2 days(SRC). 1,2-Difluoroethane's Henry's Law constant indicates that volatilization from moist soil surfaces may occur(SRC). 1,2-Difluoroethane is expected to volatilize from dry soil surfaces(SRC) based upon a vapor pressure of 616 mm Hg(3).
Occupational exposure to 1,2-difluoroethane may have occured through inhalation and dermal contact with this compound at workplaces where 1,2-difluoroethane was produced or used. (SRC)
Drug Information
... Main factor affecting fate of fluorocarbons is body fat, where they are concentrated and slowly released into blood at concn that should not cause any risk of cardiac sensitization. /Fluorocarbons/|There is a significant accumulation of fluorocarbons in brain, liver, and lung compared to blood levels, signifying a tissue distribution of fluorocarbons similar to that of chloroform. /Fluorocarbons/|Abosrption of fluorocarbons is much lower after oral ingestion (35-48 times) than after inhalation. ... The lung generally has the highest fluorocarbon concentrations on autopsy. /Fluorocarbons/|Although fluorocarbons cause cardiac sensitization in certain animal species, rapid elimination prevents the development of cardiotoxic concentrations from aerosol bronchodilator use except at exceedingly high doses (12 to 24 doses in 2 minutes). /Fluorocarbons/|Fluorocarbon compounds are lipid-soluble and thus are generally well absorbed through lung. Absorption after ingestion is 35 to 48 times lower than after inhalation ... Fluorocarbons are eliminated by way of lung. /Fluorocarbon compounds/
If the diagnosis of solvent abuse is suspected it can be confirmed by biochemical examination of the blood or urine. Emergency treatment is supportive and includes decontamination, oxygen, and any specific therapy required in a particular case such as antiarrhythmics or anticonvulsants. A few patients may require intermittent positive-pressure ventilation, dialysis, or treatment for hepatic failure. /Solvent abuse/|... In persons who are intoxicated with fluorocarbons, steps can be taken to lessen the risk of arrhythmias. ... Before evaluation at the hospital, patients should be advised to avoid strenuous exercise. In the hospital, patients can be placed in a quiet, nonthreatening environment and sedated if necessary. If hypoxic, oxygen should be administered and metabolic abnormalities corrected. Sympathomimetic drugs should be avoided. Ventricular arrhythmias are best treated with beta-blocking agents. /Fluorocarbons/|Victims of Freon inhalation require management for hypoxic, CNS anesthetic, and cardiac symptoms. Patients must be removed from the exposure environment, and high-flow supplemental oxygen should be utilized. The respiratory system should be evaluated for injury, aspiration, or pulmonary edema and treated appropriately. CNS findings should be treated supportively. A calm environment with no physical exertion is imperative to avoid increasing endogenous adrenegic levels. Exogenous adrenergic drugs must not be used to avoid inducing sensitized myocardial dysrhythmias. Atropine is ineffective in treating bradyarrhythmias. For ventricular dysrhythmias, diphenylhydantoin and countershock may be effective. Cryogenic dermal injuries should be treated by water bath rewarming at 40 to 42 °C until vasodilatory flush has returned. Elevation of the limb and standard frostbite management with late surgical debridement should be utilized. Ocular exposure requires irrigation and slit-lamp evaluation for injury. /Freons/|Immediate first aid: Ensure that adequate decontamination has been carried out. If patient is not breathing, start artificial respiration, preferably with a demand-valve resuscitator, bag-valve-mask device, or pocket mask, as trained. Perform CPR as necessary. Immediately flush contaminated eyes with gently flowing water. Do not induce vomiting. If vomiting occurs, lean patient forward or place on left side (head-down position, if possible) to maintain an open airway and prevent aspiration. Keep patient quiet and maintain normal body temperature. Obtain medical attention. /Chlorinated fluorocarbons (CFCs) and related compounds/|For more Antidote and Emergency Treatment (Complete) data for 1,2-Difluoroethane (8 total), please visit the HSDB record page.
/OTHER TOXICITY INFORMATION/ Excessive skin contact with liq fluorocarbons should be minimized to prevent defatting of skin ... /Fluorocarbons/|/OTHER TOXICITY INFORMATION/ ... The combination of fluorocarbon with a sympathomimetic bronchodilator is potentially dangerous for the treatment of bronchial asthma. For the same reason, sympathomimetic drugs are contraindicated in cardiac resuscitation of patients suffering from fluorocarbon poisoning. /Fluorocarbon poisoning/|/OTHER TOXICITY INFORMATION/ Fluorocarbon vapors are 4 to 5 times heavier than air. Thus high concn tend to accumulate in low-lying areas, resulting in hazard of inhalation of concentrated vapors, which may be fatal. /Fluorocarbons/|/OTHER TOXICITY INFORMATION/ Clinical pathologists exposed to fluorocarbons in the prepn of frozen tissue sections have been seen to develop coronary heart disease. /Fluorocarbons/|/OTHER TOXICITY INFORMATION/ Freons are toxic to humans by several mechanisms. Inhaled fluorocarbons sensitized the myocardium to catecholamines, frequently resulting in lethal ventricular arrhythmias. Because they are gases heavier than air, fluorocarbons can displace atmospheric oxygen, thus resulting in asphyxiation. These compounds also have a central nervous system (CNS) anesthetic effect analogous to a structurally similar general anesthetic, halothane. Pressurized refrigerant or liquid fluorocarbons with a low boiling point have a cryogenic effect on exposed tissues, causing frostbite, laryngeal or pulmonary edema, and gastrointestinal perforation. Certain fluorocarbons degrade at high temperatures into toxic products of chlorine, hydrofluoric acid, or phosgene gases. /Freons/
1,2-Difluoroethane Use and Manufacturing
Manufacturing processes use hydrofluoric acid from fluorospar in production of all fluorocarbons. Some processes use carbon tetrachloride from carbon disulfide, or as co-product of perchloroethylene and chlorination of propylene, or chloroform from chlorination of methanol. The major hazards relate primarily to the inadvertent release of hydrofluoric acid or carbon tetrachloride, rather than to the manufactured fluorocarbons. /Fluorocarbons/
Mechanical vapor compression systems use fluorocarbons for refrigeration and air conditioning and account for ... majority of refrigeration capability in US ... Fluorocarbons are used as refrigerants in home appliances, mobile air conditioning units, retail food refrigeration systems and ... chillers. /Fluorocarbons/
(1984) 1.36x10+11 g (est) /CFC-13, -113, -114, -115, Fluorinated monomers and specialities/
Refrigerants, 39%; foam blowing agents, 17%; solvents, 14%; fluoropolymers, 14%; sterilant gas, 2%; aerosol propellants, 2%; food freezant, 1%; other, 8%; exports, 3% (1985) /Fluorocarbons/
GAS CHROMATOGRAPHIC METHOD FOR DETERMINING FLUOROCARBONS IN AIR IS DESCRIBED. CONCN IN AIR ARE DETERMINED DIRECTLY. /FLUOROCARBONS/|GAS CHROMATOGRAPHIC METHOD FOR MEASURING HALOCARBONS IN AMBIENT AIR SAMPLES IS PRESENTED. /HALOCARBONS/|FLUOROCARBONS IN AIR OF WORKING AREA & IN EXHALED AIR CAN BE ANALYZED BY IR SPECTROMETRY. /FLUOROCARBONS/|GAS CHROMATOGRAPHIC METHOD IS PRESENTED FOR FREONS. /FREONS/|A GAS CHROMATOGRAPHIC PROCEDURE FOR DETERMINING ATMOSPHERIC LEVELS OF FLUOROCARBONS IS DESCRIBED. COLUMN IS TEMP PROGRAMMED TO SEPARATE HALOGENATED COMPONENTS WHILE MAINTAINING SHORT RETENTION TIMES FOR EACH COMPONENT. FREON 113 INCL. /FLUOROCARBONS/
GAS CHROMATOGRAPHIC METHOD FOR DETERMINING FLUOROCARBONS IS DESCRIBED. CONCN IN BODY FLUIDS ARE DETERMINED BY MEANS OF HEAD SPACE ANALYSIS. /FLUOROCARBONS/|FLUOROCARBON DETERMINATION IN BLOOD: GAS CHROMATOGRAPHY WITH ELECTRON CAPTURE DETECTION. /FLUOROCARBONS/
Computed Properties
Molecular Weight:66.05
XLogP3:0.9
Hydrogen Bond Acceptor Count:2
Rotatable Bond Count:1
Exact Mass:66.02810645
Monoisotopic Mass:66.02810645
Heavy Atom Count:4
Complexity:6
Covalently-Bonded Unit Count:1
Compound Is Canonicalized:Yes
Learn More Other Chemicals
-
4-(Trifluoromethyl)benzeneacetic acid
32857-62-8
-
3-BROMO-2-FLUORO-6-PICOLINE
375368-78-8
-
2,4-Diisopropylphenol
2934-05-6
-
1,2-Diiodotetrafluoroethane Formula
354-65-4
-
6-METHYL-2-PIPERIDINECARBOXYLICACID Formula
99571-58-1
-
2,4-Dichloro-6-picoline Formula
42779-56-6
-
1-BROMO-1H,1H,2H,2H-PERFLUORODECANE Structure
21652-57-3
-
Perfluorohexanoic acid Structure
307-24-4
-
What is 6-chloro-2-naphthalenethiol
392330-26-6
-
What is 2,3-Dichloro-5-nitropyridine
22353-40-8




