6-Methyl-2-(methylthio)pyrimidin-4-ol
-
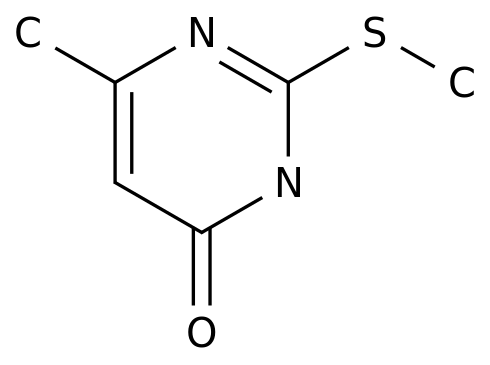
6-Methyl-2-(methylthio)pyrimidin-4-ol
structure -
-
CAS No:
6328-58-1
-
Formula:
C6H8N2OS
-
Chemical Name:
6-Methyl-2-(methylthio)pyrimidin-4-ol
-
Synonyms:
NSC37678;NSC43815;ST074533;6328-58-1;NSC193525;ST5114026;ZINC00039515;ZINC03864720;ZINC05182359;Enamine_001117
- Categories:
-
CAS No:
6-Methyl-2-(methylthio)pyrimidin-4-ol Basic Attributes
156.21
156.035736
193525|43815|37678
DTXSID50284570
2933599090
Safety Information
3
22-37/38-41
26-39-24/25
Xn
P261, P264, P270, P271, P280, P301+P312, P302+P352, P304+P340, P305+P351+P338, P310, P312, P321, P330, P332+P313, P362, P403+P233, P405, P501
H302
6-Methyl-2-(methylthio)pyrimidin-4-ol Use and Manufacturing
(EV-AO5743-001) [00128] To water (500 mL) was added NaOH (97percent, 15.7 g, 381 mmol) and the suspension stirred at r.t. for 10 mins. 6-methyl-2-sulfanylidene-l, 2, 3, 4-tetrahydropyrimidin-4-one (98percent, 53.5 g, 369mmol) was added and the mixture stirred until fully dissolved for 10 mins. lodomethane (28.99 mL, 461 mmol) was added dropwise and the mixture stirred at r.t. for 4 h. The colorless solid was filtered, washed with ice cold water (2 x 100 mL) and dried under vacuum at 60 °C to afford the title compound (57 g, 98percent) as a colorless solid. [00129] Method A: LC-MS m/z = 156.9 [M + H]To a solution of 2-methyl-2-thiopseudourea sulfate (Aldrich, 58.74 g, 0.422 mol) in water (1000 mL) were added sodium carbonate (81.44 g, 0.768 mol) and ethyl acetoacetate (50 g, 0.384 mol) at room temperature. The reaction mixture was stirred overnight. After neutralizing to pH = 8, the solid was collected through filtration followed by drying under vacuum overnight to afford 6-methyl-2-(methylthio)pyrimidin-4(3H)-one (57.2 g, 95percent yield) of product. Synthesis 1; 2-amino-8-ethyl-4-methyl-6-(li/-pyrazol-3-yl)pyrido[2, 3-d]pyriniidin-7(8//)-one; [00235] To a solution of 2-methyl-2-thiopseudourea sulfate (Aldrich, 58.74 g, 0.422 mol) in water (1000 mL) were added sodium carbonate (81.44 g, 0.768 mol) and ethyl acetoacetate (50 g, 0.384 mol) at room temperature. The reaction mixture was stirred overnight. After neutralizing to pH 8, the solid was collected through filtration followed by drying under vacuum overnight to afford 6-methyl-2-(methylthio)pyrimidin-4(3H)-one (57.2 g, 95percent yield) of product. Intermediate 1; 2-amino-8-ethyl-4-methyl-6-(lJϊ-pyrazol-3-yl)pyrido[2, 3-d]pyrimidm-7(8To a solution of the pyrimidine (324a; 522.3 g) in DMSO(5 L), potassium carbonate (535.6 g) followed by iodomethane (245 ml) were added while maintaining a reaction temperature of 22-25 C (dry ice/acetone bath). When the addition was complete the reaction was allowed to stir at room temperature overnight. Ice (7 L) and water (13 L) wereadded to the reaction. Afier 0.5 h., the mixture was filtered and the solid washed with cold water, cold acetonitrile and cold ether to give the methyl sulfide (324b; 95.7 g). To the filtrate was added 50percent aq. HC1 (300 ml) while cooled in a dry ice/acetone bath. Afier stirring for 5 mm, thewhite solid was collected, Afier washing with cold water, acetonitrile and ether a thrther portion of the methylsulfide (324b; 361.2 g) was recovered.
Computed Properties
Molecular Weight:156.21
XLogP3:0.6
Hydrogen Bond Donor Count:1
Hydrogen Bond Acceptor Count:3
Rotatable Bond Count:1
Exact Mass:156.03573406
Monoisotopic Mass:156.03573406
Topological Polar Surface Area:66.8
Heavy Atom Count:10
Complexity:220
Covalently-Bonded Unit Count:1
Compound Is Canonicalized:Yes
Recommended Suppliers of 6-Methyl-2-(methylthio)pyrimidin-4-ol
-
![China]() CN
CN
4 YRS
Business licensedTrader Supplier of Intermediates,Building blocks,API,Silicones,Peptides,Lab chemicals,Biochemicals,Pharmaceuticals,Screening Compounds,Food Additives
Learn More Other Chemicals
-
(2E)-3-(1-METHYL-1H-PYRROL-2-YL)ACRYLIC ACID
51485-76-8
-
Benadryl N-oxide hydrochloride
13168-00-8
-
6-CHLORO-3-IODO-IMIDAZO[1,2-A]PYRIDINE
885275-59-2
-
(2-Bromophenyl)diphenylphosphine Formula
62336-24-7
-
1-Morpholinocyclopentene Formula
936-52-7
-
4-[2-(Boc-amino)ethoxy]-benzoic acid Formula
168892-66-8
-
3-amino-5-bromopyridine-2-carboxylic acid Structure
870997-85-6
-
2-Amino-6-methylpyridine Structure
1824-81-3
-
What is 3-Bromo-2-methylthiophene
30319-05-2
-
What is THIOPHEN-2-YLMETHYL-PHOSPHONICACIDDIETHYLESTER
2026-42-8
6-Methyl-2-(methylthio)pyrimidin-4-ol
SDSRequest for Quotation





