2-Hydroxy-4-nitrobenzaldehyde
-
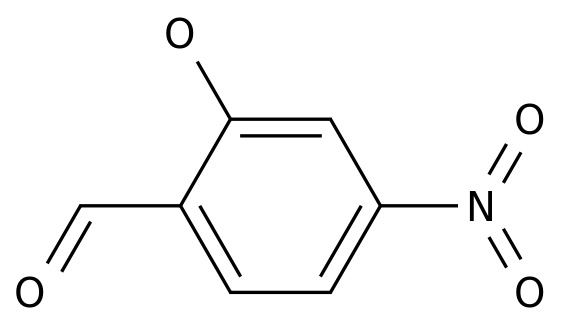
2-Hydroxy-4-nitrobenzaldehyde
structure -
-
CAS No:
2460-58-4
-
Formula:
C7H5NO4
-
Chemical Name:
2-Hydroxy-4-nitrobenzaldehyde
-
Synonyms:
Benzaldehyde,2-hydroxy-4-nitro-;Salicylaldehyde,4-nitro-;2-Hydroxy-4-nitrobenzaldehyde;4-Nitrosalicylaldehyde;NSC 82622
- Categories:
-
CAS No:
2-Hydroxy-4-nitrobenzaldehyde Basic Attributes
167.12
167.12
219-549-4
82622
DTXSID80179349
2913000090
Safety Information
36/37
|Warning|H302 (100%): Harmful if swallowed [Warning Acute toxicity, oral]|P261, P264, P270, P271, P280, P301+P312, P302+P352, P304+P340, P305+P351+P338, P312, P321, P330, P332+P313, P337+P313, P362, P403+P233, P405, and P501|The GHS information provided by 1 company from 1 notification to the ECHA C&L Inventory.
2-Hydroxy-4-nitrobenzaldehyde Use and Manufacturing
General procedure: In a 10-mL round-bottom flask equipped with O2-filledballoon (~1 atm.), a mixture of alcohol (1 mmol), absolute ethanol (3 mL) and 10 mg of catalyst 4 (3.2 molpercent) was stirred at room temperature. After the completion of the reactions that monitored by thin layer chromatography(TLC), the catalyst was filtered off and the reaction mixture was extracted to CH2Cl2and were purified with silica gel plate chromatography (Scheme 2). The oxidation of primary alcohols proceeds to aldehyde stage and was not observed any carboxylic acid product identified by TLC.The reactions also were performed in the absence of catalyst or in the absence of oxidant in the same conditions.The compound 2- (hydroxymethyl) -5-nitrophenol (13 g, 77 mmol) and manganese dioxide (26.7 g, 308 mmol) were added to ethyl acetate (250 mL)Heated to 80 ° C for 16 h.filter, The filtrate was concentrated, The crude product was isolated by column chromatography (petroleum ether / ethyl acetate = 1: 3, v / v) to give 2-hydroxy-4-nitrobenzaldehyde (10.5 g)The yield was 81.7percent.Step 2; The alcohol 5-2 from step 1 (69.6 g, 0.41 mole) was charged in a 5 L 3-necked flaskequipped with a reflux condenser and mechanical stirrer. Chloroform (2.5 L) wasadded and the mixture heated slowly. Activated MnOCompound 19 (4.28 g, 25.3 mmol), active MnO2 (4.43 g, 50.71 mmol) was added to chloroform (80 mL)The reaction mixture was refluxed for 20 hours, cooled to room temperature, filtered through celite to remove the solid, washed with chloroform and the filtrate was concentrated. The residue was purified by silica gel column chromatography (elution: petroleum ether: ethyl acetate = 10: 1) 2.62 g of compound 20 in a yield of 62percent.General procedure: In a 10-mL round-bottom flask equipped with O2-filledballoon (~1 atm.), a mixture of alcohol (1 mmol), absolute ethanol (3 mL) and 10 mg of catalyst 4 (3.2 molpercent) was stirred at room temperature. After the completion of the reactions that monitored by thin layer chromatography(TLC), the catalyst was filtered off and the reaction mixture was extracted to CH2Cl2and were purified with silica gel plate chromatography (Scheme 2). The oxidation of primary alcohols proceeds to aldehyde stage and was not observed any carboxylic acid product identified by TLC.The reactions also were performed in the absence of catalyst or in the absence of oxidant in the same conditions.General procedure: Alcohol (10.0 mmol) and the catalyst 9 (40 mg, 0.5 mol percent Mn)were added to 1.5mL H2O2 37percent (10 mL round bottom flask). Thereaction was stirred at room temperature in air atmosphere forsufficient time. Progress of the reaction and product selectivitywere monitored by GC. After completion of the reaction, the catalyst9 was separated by an external magnet. The product was filteroff, washed with deionized water (3 x 5 mL), dried into oven(50 °C) and then isolated. Silica-gel plate chromatography utilizedfor the case of benzaldehyde purification.The compound 2- (hydroxymethyl) -5-nitrophenol (13 g, 77 mmol) and manganese dioxide (26.7 g, 308 mmol) were added to ethyl acetate (250 mL)Heated to 80 ° C for 16 h.filter, The filtrate was concentrated, The crude product was isolated by column chromatography (petroleum ether / ethyl acetate = 1: 3, v / v) to give Step 2; The alcohol 5-2 from step 1 (69.6 g, 0.41 mole) was charged in a 5 L 3-necked flaskequipped with a reflux condenser and mechanical stirrer. Chloroform (2.5 L) wasadded and the mixture heated slowly. Activated MnO2 (85percent, 252 g, 2.46 moles, 6equivalents) was added in small portions over 15 min, during which time, thetemperature reached 55-58°C. Reflux was maintained for 1 h, after which TLCanalysis indicated complete conversion to the aldehyde. The reaction mixture wascooled to room temperature and filtered through celite using CHCI3 for washings. Theyellow filtrate was concentrated under reduced pressure to give aldehyde 5-3 as a ayellow solid that was dried under vacuum (52.11 g, 75percent yield).5 g of the compound 1 (molecular formula: C7H5NO4) (30.65 mmol) was dissolved in ethyl acetate, and 20 g of MnO 2 (230 mmol) was further added thereto, and the reaction liquid was heated to about 80 ° C, refluxed, and stirred for 6 hours, and the reaction was completed. After the reaction is completed, The reaction solution was cooled to room temperature, and ethyl acetate was used as an eluent. The first purification of MnO2 was carried out on a silica gel column to obtain a solid compound, which was washed with petroleum ether and ethyl acetate in a volume ratio of 3:1. The mixture was stripped and subjected to secondary purification using a silica gel column to give 3.2 g of a yellow solid (yield: 64.7percent).Compound 19 (4.28 g, 25.3 mmol), active MnO2 (4.43 g, 50.71 mmol) was added to chloroform (80 mL)The reaction mixture was refluxed for 20 hours, cooled to room temperature, filtered through celite to remove the solid, washed with chloroform and the filtrate was concentrated. The residue was purified by silica gel column chromatography (elution: petroleum ether: ethyl acetate = 10: 1) 2.62 g of compound 20 in a yield of 62percent.
Computed Properties
Molecular Weight:167.12
XLogP3:2
Hydrogen Bond Donor Count:1
Hydrogen Bond Acceptor Count:4
Rotatable Bond Count:1
Exact Mass:167.02185764
Monoisotopic Mass:167.02185764
Topological Polar Surface Area:83.1
Heavy Atom Count:12
Complexity:188
Covalently-Bonded Unit Count:1
Compound Is Canonicalized:Yes
Recommended Suppliers of 2-Hydroxy-4-nitrobenzaldehyde
-
![China]() CN
CN
4 YRS
Business licensed Certified factoryManufactory Supplier of fine chemicals,pharmaceutical materials
Learn More Other Chemicals
-
Debilone
26808-51-5
-
D2 C20 TRIAROMATIC STERANE
205529-79-9
-
D-[1,3-13C2]RIBOSE
478511-79-4
-
DECAHYDRO-2-(IODOMETHYL)-NAPHTHALENE Formula
409112-76-1
-
3-DEOXY-3-FLUORO-D-GALACTOSE Formula
52904-86-6
-
3-DEOXY-3-FLUORO-D-ALLOSE Formula
99605-33-1
-
3-Deoxy-D-arabinose Structure
55658-87-2
-
3-DEOXY-3-FLUORO-D-MANNOSE Structure
87764-46-3
-
What is 3-Deoxy-L-arabinose
41107-43-1
-
What is 3-DEOXY-3-FLUORO-D-GLUCITOL
34339-82-7





