2-Chlorophenylboronicacid
-
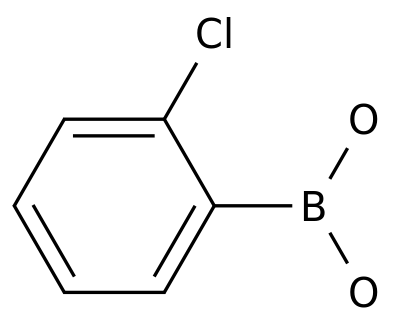
2-Chlorophenylboronicacid
structure -
-
CAS No:
3900-89-8
-
Formula:
C6H6BClO2
-
Chemical Name:
2-Chlorophenylboronicacid
-
Synonyms:
AKOSBRN-0009;RARECHEMAHPB0177;2-Chlorophenylboroni;2-Chlorophenylboronic;2-chlorobenzeneboronic;Chlorophenylboronicacid;2-CHLORPHENYLBORONSAEURE;2-CHLOROPHENYLBORNICACID;2-CHLOROPHENYLBORONICAICD;O-CHLOROPHENYLBORONICACID
- Categories:
-
CAS No:
Characteristics
40.5
0.01980
white crystalline powder
1.3±0.1 g/cm3
92-102 °C(lit.)
306.3°C at 760 mmHg
139.0±28.4 °C
1.558
0-6ºC
Safety Information
NONH for all modes of transport
3
R36/37/38
S37/39-S26
Xi:Irritant
P261, P264, P270, P271, P280, P301+P312, P302+P352, P304+P340, P305+P351+P338, P312, P321, P330, P332+P313, P337+P313, P362, P403+P233, P405, P501
H302
2-Chlorophenylboronicacid Use and Manufacturing
Procedure (f):n-BuLi (2.5 M in n-Hexane, 2.5 ml, 1.2 eq., 6.3 mmol) was added dropwise to a solution of Bromo chloro benzene (I) (ig, 0.6 ml, 5.2 mmol) and Triisopropylborate (1.44 ml, 1.2 eq., 6.27 mmol) in Toluene and THF (4:1, 10 ml)under nitrogen at -7 0°C over 1 hour. The reaction mixture was stirred for an additional 0.5 hour while the temperature was held at -70°C. The reaction mixture was allowed to warm to -20°C, before a 2 N HC1 solution (5 ml) was added to the reaction mixture. When the reaction mixture reached room temperature, it was extracted with Dichloromethane. Combined organic phase was dried and evaporatedto give a white solid, which was recrystallized from MeCN with a yield of 98percent (800mg).General procedure: Under an argon atmosphere a solution of the appropriate bromobenzene (1 equivalent) dissolved in anhydrous THF (approximately 30 mL per mmol bromobenzene) is cooled to -78 °C using a nitrogen-ethanol-bath. A solution of 2.3 equivalents of n-butyllithium in hexane is added drop wise keeping the temperature below -78 °C. After completion the mixture is stirred for one hour at this temperature. Then 1.5 equivalents of trimethyl borate are added slowly and the reaction mixture is stirred at -78 °C for another hour. The cooling bath is then removed, the reaction mixture is stirred until room temperature is reached and quenched with a saturated solution of ammonium chloride. THF and the major part of the water is removed under reduced pressure, the residue is laced with 3M hydrochloric acid until a pH of 3 is reached. After extraction with DCM (3 x) the organic phases are collected, washed with brine, dried over sodium sulphate and filtered. DCM is removed under reduced pressure, the resulting solid is washed first with ice cold water and then with PE and dried.A.
suzuki reaction
Computed Properties
Molecular Weight:156.38
Hydrogen Bond Donor Count:2
Hydrogen Bond Acceptor Count:2
Rotatable Bond Count:1
Exact Mass:156.0149373
Monoisotopic Mass:156.0149373
Topological Polar Surface Area:40.5
Heavy Atom Count:10
Complexity:110
Covalently-Bonded Unit Count:1
Compound Is Canonicalized:Yes
Recommended Suppliers of 2-Chlorophenylboronicacid
-
![China]() CN
CN
3 YRS
Business licensed Certified factoryManufactory Supplier of Chemical Pesticides,Food Additives,Agrochemicals,Active Pharm Ingredients,Flavors and Fragrances,Chemical Catalyst,Chemical Materials,Chem&Pharm Intermediates,Organic Intermediates,Feed Additive -
![China]() CN
CN
4 YRS
Business licensed Certified factoryManufactory Supplier of fine chemicals,pharmaceutical materials -
![China]() CN
CN
5 YRS
Business licensed Certified factoryManufactory Supplier of nano silver solution,precious metal catalyst,OLED materialInquiryCAS No.: 3900-89-8Grade: Industrial GradeContent: 99.5%
Learn More Other Chemicals
-
Argininosuccinicacidhydratedisodiumsalt
918149-29-8
-
ARISTOLOCHIC ACID D(RG)(PLEASE CALL)
17413-38-6
-
1(10)-Aristolen-2-one
28398-06-3
-
ASISCHEM Z06232 Formula
85209-63-8
-
Asukamycin Formula
61116-33-4
-
7-Azabicyclo[2.2.1]heptane-1-carboxamide,N-methyl-(9CI) Formula
783298-25-9
-
1-Azabicyclo[3.2.2]nonan-3-one(9CI) Structure
473795-47-0
-
2-Azabicyclo[2.2.1]heptan-3-one,6,6-difluoro-(9CI) Structure
262280-03-5
-
What is 3′-Azido-3′-deoxy-5′-O-(triphenylmethyl)thymidine
29706-84-1
-
What is 3'-AZIDO-2'-3'-DIDEOXYGUANOSINE
66323-46-4





