2-Chloro-4-nitroaniline
-
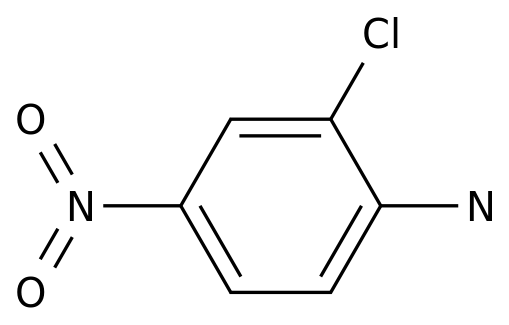
2-Chloro-4-nitroaniline
structure -
-
CAS No:
121-87-9
-
Formula:
C6H5ClN2O2
-
Chemical Name:
2-Chloro-4-nitroaniline
-
Synonyms:
Benzenamine,2-chloro-4-nitro-;Aniline,2-chloro-4-nitro-;2-Chloro-4-nitrobenzenamine;o-Chloro-p-nitroaniline;2-Chloro-4-nitroaniline;OCPNA;1-Amino-2-chloro-4-nitrobenzene;2-Chloro-4-nitrophenylamine;4-Nitro-2-chloroaniline;NSC 3548
- Categories:
-
CAS No:
Description
yellow crystalline powderYellow crystalline powder.
2-chloro-4-nitroaniline is a yellow crystalline powder. (NTP, 1992)
2-chloro-4-nitroaniline is a yellow crystalline powder. (NTP, 1992)
2-Chloro-4-nitroaniline Basic Attributes
172.57
172.57
638657
204-502-2
XSA3ZX337B
3548
2237
DTXSID4021973
Yellow needles from water
2921 42 00
Characteristics
71.8
2.3
2-chloro-4-nitroaniline is a yellow crystalline powder. (NTP, 1992)
<1 g/cm3
108 °C
>200 °C
205 °C
1.6460 (estimate)
H2O: 0.23 g/L (20 ºC)
Store below +30°C.
Vapour pressure, Pa at 25°C: 0.00046
Oral-Rat LD50:6430 mg/kg; Oral-Mouse LD50:1250 mg/kg
Open flame is flammable; react with oxidant; high heat or contact with acid releases chloride and nitrogen oxide gas
9.54e-09 atm-m3/mole|Henry's Law constant = 0.54X10-9 atm-cu m/mol at 25 °C
pKa = -0.94 (conjugate acid)
Hydroxyl radical reaction rate constant = 3.93X10-12 cu cm/molec-sec at 25 °C (est)
Insoluble in water.
Nitro, Nitroso, Nitrate, and Nitrite Compounds, Organic
2-CHLORO-4-NITROANILINE can react with oxidizing materials. (NTP, 1992)
Safety Information
III
6.1(b)
UN 2237 6.1/PG 3
2
22-51/53
22-24-61
BX1400000
Xn,N
The warehouse is ventilated at low temperature and dry; stored separately from oxidants, acids and food additives
Stable. Incompatible with strong oxidizing agents, strong bases, strong acids.
P273
H302-H411
SRP: The most favorable course of action is to use an alternative chemical product with less inherent propensity for occupational exposure or environmental contamination. Recycle any unused portion of the material for its approved use or return it to the manufacturer or supplier. Ultimate disposal of the chemical must consider: the material's impact on air quality; potential migration in soil or water; effects on animal, aquatic, and plant life; and conformance with environmental and public health regulations.
The substance decomposes on burning producing toxic and corrosive gases, including nitrogen oxides.
European Chemicals Bureau; IUCLID Dataset, 2-Chloro-4-nitroaniline (CAS # 121-87-9) . Describes information supplied to the EU by European industries as of 2000. Available from the Database Query page at: http://ecb.jrc.it/esis/esis.php as of October 1, 2007.
Excerpt from ERG Guide 153 [Substances - Toxic and/or Corrosive (Combustible)]: Combustible material: may burn but does not ignite readily. When heated, vapors may form explosive mixtures with air: indoors, outdoors and sewers explosion hazards. Those substances designated with a (P) may polymerize explosively when heated or involved in a fire. Contact with metals may evolve flammable hydrogen gas. Containers may explode when heated. Runoff may pollute waterways. Substance may be transported in a molten form. (ERG, 2016)
|Warning|H302: Harmful if swallowed [Warning Acute toxicity, oral]|P264, P270, P273, P301+P312, P330, P391, and P501|H302 (63.12%): Harmful if swallowed [Warning Acute toxicity, oral]|P264, P270, P273, P280, P301+P312, P302+P352, P312, P322, P330, P363, P391, and P501|Aggregated GHS information provided by 141 companies from 3 notifications to the ECHA C&L Inventory. Each notification may be associated with multiple companies.|Not Classified
Excerpt from ERG Guide 153 [Substances - Toxic and/or Corrosive (Combustible)]: As an immediate precautionary measure, isolate spill or leak area in all directions for at least 50 meters (150 feet) for liquids and at least 25 meters (75 feet) for solids. SPILL: Increase, in the downwind direction, as necessary, the isolation distance shown above. FIRE: If tank, rail car or tank truck is involved in a fire, ISOLATE for 800 meters (1/2 mile) in all directions; also, consider initial evacuation for 800 meters (1/2 mile) in all directions. (ERG, 2016)
SMALL SPILLS AND LEAKAGE: If you spill this chemical, use absorbent paper to pick up all liquid spill material. Your contaminated clothing and absorbent paper should be sealed in a vapor-tight plastic bag for eventual disposal. Solvent wash all contaminated surfaces with alcohol followed by washing with a strong soap and water solution. Do not reenter the contaminated area until the Safety Officer (or other responsible person) has verified that the area has been properly cleaned. STORAGE PRECAUTIONS: You should store this material in a refrigerator. (NTP, 1992)
MINIMUM PROTECTIVE CLOTHING: When working with this chemical, you should wear an impervious full-body suit equipped with an air line respirator or a self-contained breathing apparatus. RECOMMENDED RESPIRATOR: Where the neat test chemical is weighed and diluted, wear a NIOSH-approved half face respirator equipped with an organic vapor/acid gas cartridge (specific for organic vapors, HCl, acid gas and SO2) with a dust/mist filter. RECOMMENDED GLOVE MATERIALS: Permeation data indicate that butyl rubber gloves may provide protection to contact with this compound. Butyl rubber over latex gloves is recommended. However, if this chemical makes direct contact with your gloves, or if a tear, hole or puncture develops, remove them at once. (NTP, 1992)|Local exhaust or breathing protection. Protective gloves. Safety spectacles.
Powder, alcohol-resistant foam, water spray, carbon dioxide.
Sweep spilled substance into containers; if appropriate, moisten first to prevent dusting. Carefully collect remainder, then remove to safe place. (Extra personal protection: P2 filter respirator for harmful particles). Do NOT let this chemical enter the environment.
PREVENT DISPERSION OF DUST! Do not eat, drink, or smoke during work. Wash hands before eating.
Do not transport with food and feedstuffs. Marine pollutant.
Toxicity
moderately toxic
LD50 Mouse oral 1250 mg/kg bw|LD50 Rat oral 6430 mg/kg bw
/AQUATIC SPECIES/ The Dreissena-Monitor is a biological early warning system for the continuous monitoring of river water quality, based on the valve movements of two groups of 42 zebra mussels (Dreissena polymorpha). Laboratory experiments with ... 2-chloro-4-nitro-aniline were conducted in combination with suspended particles (a mixture of stinging nettle powder, bentonite, and quartz powder). An increase of suspended particles up to a nominal concentration of 540 mg/L within 5 min did not evoke any reactions by the mussels significantly different from normal. The distribution between water and solids was analyzed for ... 2-chloro-4-nitroaniline, with the result that /it/ ... did not bind to the particles at all. The behavior of the zebra mussels revealed that the detection of 2-chloro-4-nitro-aniline was not affected by the presence of suspended matter. ...
Routine checking of lips, tongue and nail beds of exposed personnel for signs of cyanosis. /Protect/ from exposure those individuals with anemia, cardiovascular or pulmonary diseases.
2-Chloro-4-nitroaniline's production and use as an intermediate in the manufacture of both disperse and basic dyes(1,2) and in the manufacture of niclosamid, a molluscicide(3), may result in its release to the environment through various waste streams(SRC).
TERRESTRIAL FATE: Based on a classification scheme(1), an estimated Koc value of 350(SRC), determined from a log Kow of 2.14(2) and a regression-derived equation(3), indicates that 2-chloro-4-nitroaniline is expected to have moderate mobility in soil(SRC). Volatilization of 2-chloro-4-nitroaniline from moist soil surfaces is not expected to be an important fate process(SRC) given a Henry's Law constant of 9.5X10-9 atm-cu m/mole(4). 2-Chloro-4-nitroaniline is not expected to volatilize from dry soil surfaces(SRC) based upon an estimated vapor pressure of 4.8X10-4 mm Hg(SRC), determined from a fragment constant method(5). 2-Chloro-4-nitroaniline is regarded as non-biodegradable in the aquatic environment as well as in communal and industrial sewage treatment plants(6); therefore, biodegradation is not an important environmental fate process in soil(SRC).|AQUATIC FATE: Based on a classification scheme(1), an estimated Koc value of 350(SRC), determined from a log Kow of 2.14(2) and a regression-derived equation(3), indicates that 2-chloro-4-nitroaniline is expected to adsorb to suspended solids and sediment(SRC). Volatilization from water surfaces is not expected(3) based upon a Henry's Law constant of 9.5X10-9 atm-cu m/mole(4). The stability of 2-chloro-4-nitroaniline in standardized aqueous medium was monitored at room temperature; a 12% loss of 2-chloro-4-nitroaniline was noted over a 8 day period due to either volatilization or abiotic transformation of this compound(5). According to a classification scheme(6), an estimated BCF of 9(SRC), from its log Kow(2) and a regression-derived equation(7), suggests the potential for bioconcentration in aquatic organisms is low(SRC). 2-Chloro-4-nitroaniline is regarded as non-biodegradable in the aquatic environment as well as in communal and industrial sewage treatment plants(8); therefore, biodegradation is not an important environmental fate process in water(SRC).|ATMOSPHERIC FATE: According to a model of gas/particle partitioning of semivolatile organic compounds in the atmosphere(1), 2-chloro-4-nitroaniline, which has an estimated vapor pressure of 4.8X10-4 mm Hg at 25 °C(SRC), determined from a fragment constant method(2), is expected to exist solely as a vapor in the ambient atmosphere. Vapor-phase 2-chloro-4-nitroaniline is degraded in the atmosphere by reaction with photochemically-produced hydroxyl radicals(SRC); the half-life for this reaction in air is estimated to be 4 days(SRC), calculated from its rate constant of 3.9X10-12 cu cm/molecule-sec at 25 °C(SRC) that was derived using a structure estimation method(3). 2-Chloro-4-nitroaniline contains chromophores that absorb at wavelengths >290 nm(4) and therefore may be susceptible to direct photolysis by sunlight(SRC).
The rate constant for the vapor-phase reaction of 2-chloro-4-nitroaniline with photochemically-produced hydroxyl radicals has been estimated as 3.9X10-12 cu cm/molecule-sec at 25 °C(SRC) using a structure estimation method(1). This corresponds to an atmospheric half-life of about 4 days at an atmospheric concentration of 5X10+5 hydroxyl radicals per cu cm(1). The stability of 2-chloro-4-nitroaniline in standardized aqueous medium was monitored at room temperature; a 12% loss of 2-chloro-4-nitroaniline was noted over an 8 day period due to either volatilization or abiotic transformation of this compound(2). 2-Chloro-4-nitroaniline is not expected to undergo hydrolysis in the environment due to the lack of functional groups that hydrolyze under environmental conditions(3). 2-Chloro-4-nitroaniline does contain chromophores that absorb at wavelengths >290 nm(3,4) and therefore may be susceptible to direct photolysis by sunlight(SRC).
An estimated BCF of 9 was calculated in fish for 2-chloro-4-nitroaniline(SRC), using a log Kow of 2.14(1) and a regression-derived equation(2). According to a classification scheme(3), this BCF suggests the potential for bioconcentration in aquatic organisms is low(SRC).
The Koc of 2-chloro-4-nitroaniline is estimated as 350(SRC), using a log Kow of 2.14(1) and a regression-derived equation(2). According to a classification scheme(3), this estimated Koc value suggests that 2-chloro-4-nitroaniline is expected to moderate mobility in soil. However, anilines are expected to bind strongly to humus or organic matter in soils due to the high reactivity of the aromatic amino group(4,5), suggesting that mobility may be much lower in some soils(SRC). The log Koc of 2-chloro-4-nitroaniline was measured as 2.36 in Yangtze river sediment (37.1% sand, 49.3% silt, 13.6% clay, 1.28% organic carbon, pH 7.44)(1). The log Koc was also measured as 3.63 in modified clay(6). These values correspond to Koc values of 230 and 4300, respectively(SRC).
The Henry's Law constant for 2-chloro-4-nitroaniline is 9.5X10-9 atm-cu m/mole(1). This Henry's Law constant indicates that 2-chloro-4-nitroaniline is expected to be essentially nonvolatile from water surfaces(2). 2-Chloro-4-nitroaniline is not expected to volatilize from dry soil surfaces(SRC) based upon an estimated vapor pressure of 4.8X10-4 mm Hg(SRC), determined from a fragment constant method(3).
NIOSH (NOES Survey 1981-1983) has statistically estimated that 737 workers (none of these are female) are potentially exposed to 2-chloro-4-nitroaniline in the US(1). Occupational exposure to 2-chloro-4-nitroaniline may occur through dermal contact with this compound at workplaces where 2-chloro-4-nitroaniline is produced or used(SRC).
Drug Information
As in the case of other secondary amides, phase I metabolism of niclosamide may result in a hydrolytic cleavage of the amide bond, giving rise to 5-chlorosalicylic acid and 2-chloro-4-nitroaniline as the main metabolites.
Excerpt from ERG Guide 153 [Substances - Toxic and/or Corrosive (Combustible)]: TOXIC; inhalation, ingestion or skin contact with material may cause severe injury or death. Contact with molten substance may cause severe burns to skin and eyes. Avoid any skin contact. Effects of contact or inhalation may be delayed. Fire may produce irritating, corrosive and/or toxic gases. Runoff from fire control or dilution water may be corrosive and/or toxic and cause pollution. (ERG, 2016)
EYES: First check the victim for contact lenses and remove if present. Flush victim's eyes with water or normal saline solution for 20 to 30 minutes while simultaneously calling a hospital or poison control center. Do not put any ointments, oils, or medication in the victim's eyes without specific instructions from a physician. IMMEDIATELY transport the victim after flushing eyes to a hospital even if no symptoms (such as redness or irritation) develop. SKIN: IMMEDIATELY flood affected skin with water while removing and isolating all contaminated clothing. Gently wash all affected skin areas thoroughly with soap and water. If symptoms such as redness or irritation develop, IMMEDIATELY call a physician and be prepared to transport the victim to a hospital for treatment. INHALATION: IMMEDIATELY leave the contaminated area; take deep breaths of fresh air. If symptoms (such as wheezing, coughing, shortness of breath, or burning in the mouth, throat, or chest) develop, call a physician and be prepared to transport the victim to a hospital. Provide proper respiratory protection to rescuers entering an unknown atmosphere. Whenever possible, Self-Contained Breathing Apparatus (SCBA) should be used; if not available, use a level of protection greater than or equal to that advised under Protective Clothing. INGESTION: DO NOT INDUCE VOMITING. If the victim is conscious and not convulsing, give 1 or 2 glasses of water to dilute the chemical and IMMEDIATELY call a hospital or poison control center. Be prepared to transport the victim to a hospital if advised by a physician. If the victim is convulsing or unconscious, do not give anything by mouth, ensure that the victim's airway is open and lay the victim on his/her side with the head lower than the body. DO NOT INDUCE VOMITING. IMMEDIATELY transport the victim to a hospital. (NTP, 1992)
IN ALL CASES CONSULT A DOCTOR. INHALATION: Symptoms: Blue skin. Blue lips or finger nails. Dizziness. Headache. Nausea. Shortness of breath. Confusion. Convulsions. Unconsciousness. Symptoms may be delayed. First aid: Fresh air, rest. Refer for medical attention. SKIN: Symptoms: Redness. First aid: Remove contaminated clothes. Rinse skin with plenty of water or shower. EYES: Symptoms: Redness. Pain. First aid: First rinse with plenty of water for several minutes (remove contact lenses if easily possible), then take to a doctor. INGESTION: Symptoms: Blue skin. Blue lips or finger nails. Dizziness. Headache. Nausea. Shortness of breath. Confusion. Convulsions. Unconsciousness. Symptoms may be delayed. First aid: Rest. Refer for medical attention.
2-chloro-4-nitroaniline
2-Chloro-4-nitroaniline Use and Manufacturing
Chlorination of nitroaniline with hydrochloric acid and sodium hypochlorite. The reaction product is filtered, washed and dried to obtain a finished product. Raw material consumption quota: p-nitroaniline 928kg/t, sodium hypochlorite (10%) 5600kg/t, 30% hydrochloric acid 5600kg/t.
Intermediate in manufacture of dyes.
Production volumes for non-confidential chemicals reported under the Inventory Update Rule. [Table#5397]
Benzenamine, 2-chloro-4-nitro-: ACTIVE
DETECTION OF O-CHLORO-P-NITROANILINE BY GAS CHROMATOGRAPHY WITH A 700-A HALL AND AN NPD (N-P DETECTOR) IN TREATMENT WORKS SLUDGES.|1-Amino-2-chloro-4-nitrobenzene was detected in D&C Red No. 36 as an impurity using liquid chromatography (C18 column, methanol/ammonium acetate gradient) with 254 nm detection. This method gave 100% recovery of 1-amino-2-chloro-4-nitrobenzene.|Method: EPA 8131; Procedure: gas chromatography; Analyte: 2-chloro-4-nitroaniline; Matrix: aqueous matrices; Detection Limit: 3.2 ug/L.
Computed Properties
Molecular Weight:172.57
XLogP3:2.1
Hydrogen Bond Donor Count:1
Hydrogen Bond Acceptor Count:3
Exact Mass:172.0039551
Monoisotopic Mass:172.0039551
Topological Polar Surface Area:71.8
Heavy Atom Count:11
Complexity:159
Covalently-Bonded Unit Count:1
Compound Is Canonicalized:Yes
Price Analysis
- Data: 2025-02-17
- Price: 10000.00Yuan/mt
- Change: 0
Recommended Suppliers of 2-Chloro-4-nitroaniline
-
![China]() CN
CN
1 YR
Business licensedTrader Supplier of Acrylonitrile,Sodium biselenite methylamine solution,Methylamine [anhydrous],Zinc dust,Hydrogen peroxide solution [content >8%],Triisopropyl borate,Fuming sulfuric acid,Lithium borohydride,Triethyl borate,Ethyl propionate,Propionic anhydride,Propylarsinic acid,Aniline,4-Aminobiphenyl,4-Aminophenol,2-Aminopropane,Propane,0,0-Dimethyl-S-(2,3-dihydro-5-methoxy-2-oxo-1,3,4-thiadiazol-3-ylmethyl) dithiophosphate,Natural resin paint,Epoxy resin paint,Amino resin paint,Rubber paint,Organic silicon resin,Cyclopentene,n-Butylcyclopentane,N-n-Butylaniline,3-Ethoxyaniline,Ethyl acetate,Acetic acid solution [10%< content <80%],Sodium ethoxide ethanol solution,Ethylamine aqueous solution [concentration 50%~70%],Sodium oxide,Ethyl nitrite,Sodium nitrite,Bromocyclopentane,1-Bromopropane,Bromoacetone,2-Bromoaniline,2-Nitrobromobenzene,3-Nitrobenzyl chloride,4-Nitro-2-methylaniline,4-Nitro-1,3-dimethylbenzene,Valeryl chloride,Glutaronitrile,Lead tetraoxide,Silicon tetrachloride,1,1,3,3-Tetrachloroacetone,Tetrapropylene,2-tert-Butylphenol,Tetradecanoyl chloride,Trichloroacetic acid,Trichlorotoluene,Aluminum trichloride [anhydrous],Phosphorus trichloride,Trimethylcyclohexylamine,1,2,3-Trimethylbenzene,Trimethylchlorosilane,Solvent benzene,Sodium hydroxide,Potassium hydroxide solution [content >30%],Hydrobromic acid,Methyl chloroacetate,Butyl chloroacetate,3-Chloronitrobenzene,4-Chlorotoluene,1-Chloropropane,2-Chloropropionic acid,4-Chloroaniline,2-Chloroaniline,2-Chloro-4-nitroaniline,4-Chloro-2-nitrophenol,2,2',4,4',5,5'-Hexabromodiphenyl ether,Hexamethyldisiloxane,Diethyl sulfate,Thioacetic acid,Aluminum phosphide,o-Toluenesulfonyl chloride,Phthalimide,Potassium antimony tartrate,2-Methoxyaniline,Formic acid,1-Methylpentanol,Methyltriethoxysilane,2-Methylaniline,2-Methyl-1-propanol,Toluene diisocyanate,Cyclooctene,Cyclopentane,Cyclohexanone,Cyclohexylamine,Cyclopropane,2-Furanmethanol,N,N-Diethyl-p-toluidine,N,N-Diethyl-1,3-propanediamine,Sodium 2,4-dinitro-5-methylphenolate,3,5-Dinitroaniline,2,6-Dinitroaniline,3,6-Dihydroxyphthalonitrile,3,3'-Dichlorobenzidine,2,4-Dichloroaniline,1,3-Dichlorobenzene,2,2-Dimethylpropionate,3,4-Dimethylaniline,2,5-Dimethylaniline,3,3'-Dimethyl-4,4'-diaminobiphenyl,3,4-Dimethylphenol,1,4-Dimethylbenzene,N,N-Dibutylaniline,Bromoacetic acid,Bromacetyl bromide,Isooctane,Isooctene,Natural gas [rich in methane] (only for chemical raw material use,not for fuel use),Tributyltin acetate,Diphenylphosphine dichloride,Sodium chloroacetate,Dicyclohexylamine,Valeric acid,n-Propyl mercaptan,1,3-Dichloropropane,Dichloromethane,tert-Butyl chloride,Cyclopentyl chloride,Phosphorous acid,Cyclopentanone,Trichloroacetaldehyde [stabilized],Stannous octoate,Calcium resin,Cyclohexane,n-Octane,1,2-Dichloroethane,Thionyl chloride,tert-Butyl hydroperoxide [79%< content <90%,water content >10%],Isopropenyl acetate,Sodium dithionite,Sodium dithionate,Propionyl chloride,Benzene,Hydrobromic acid,Triethylamine,Cyclopentyl chloride,Iodine trichloride,Triethyl borate,Trimethyl borate,Toluene-2,4-diisocyanate,Hydrochloric acid,Chloroform,Benzyl cyanide,Acetone,Sulfuric acid,TolueneInquiryUnit Price: $1 /MT FOBCAS No.: 121-87-9Grade: General reagentsContent: 98%
Learn More Other Chemicals
-
4,4′-(1,2-Diazenediyl)bis[N,N,N-trimethylbenzenaminium]
21704-61-0
-
N-(4-Chloro-2-methylphenyl)formamide
21787-81-5
-
Basic Blue 8
2185-87-7
-
3-tert-butyl-5-chloro-2-hydroxy-N-(2-nitrosophenyl)benzamide Formula
21889-00-9
-
1-Phenylcyclohexylamine Formula
2201-24-3
-
6-Hydroxymelatonin Formula
2208-41-5
-
Pyrrocaine Structure
2210-77-7
-
2-amino-N-(4-ethoxyphenyl)ethanesulfonamide Structure
22103-30-6
-
What is Bufeniode
22103-14-6
-
What is 4-(2-Quinolinyl)benzenamine
22191-97-5





