Benzhydrol
-
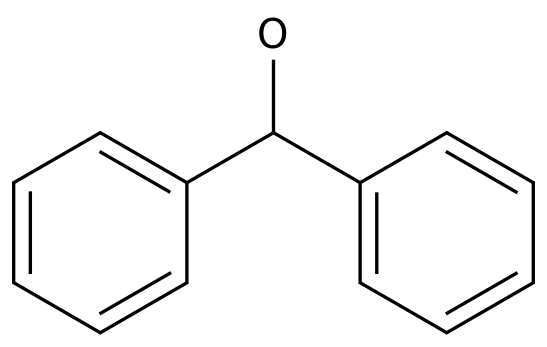
Benzhydrol
structure -
-
CAS No:
91-01-0
-
Formula:
C13H12O
-
Chemical Name:
Benzhydrol
-
Synonyms:
Benzenemethanol,α-phenyl-;Benzhydrol;α-Phenylbenzenemethanol;Diphenylmethanol;Benzohydrol;Diphenylcarbinol;Benzhydryl alcohol;Hydroxydiphenylmethane;Diphenylmethyl alcohol;α-Phenylbenzyl alcohol;NSC 32150;1,1-Diphenylmethanol
- Categories:
-
CAS No:
Description
Benzhydrol is a white to light beige crystalline solid at room temperature, and the parent member of a large class of diaryl alcohols. Benzhydrol is easily soluble in ethanol, ether, chloroform and carbon disulfide, but almost insoluble in cold crude gasoline. Its solubility in water at 20°C is only 0.5 g/L. Its molecular weight is 184.24, its melting point is 65-67 °C, its boiling point is 297-298 °C, and its density is 1.0120.
Characteristics
20.2
2.7
White to beige Crystalline Solid
1.1±0.1 g/cm3
69 °C
298 °C
160 °C
1.599
Slightly soluble in water.
Store below +30°C.
0.000076 hPa (20 °C)
LD50 orally in Rabbit: 5000 mg/kg LD50 dermal Rabbit > 5000 mg/kg
Safety Information
NONH for all modes of transport
2
36/37/38
26-36-24/25
DC7452000
Xi
Irritant
Stable. Combustible. Incompatible with strong oxidizing agents, acid chlorides, acid anhydrides, acids.
P305 + P351 + P338
H315-H319-H335
|Warning|H315 (80.33%): Causes skin irritation [Warning Skin corrosion/irritation]|P261, P264, P271, P280, P302+P352, P304+P340, P305+P351+P338, P312, P321, P332+P313, P337+P313, P362, P403+P233, P405, and P501|Aggregated GHS information provided by 61 companies from 8 notifications to the ECHA C&L Inventory. Each notification may be associated with multiple companies.
Benzhydrol Use and Manufacturing
Widely utilized in a variety of industrial applications, benzaldehyde is a chemical compound that exhibits exceptional chemical reactivity. This attribute allows it to be readily converted into benzophenone through oxidation reactions. The compounds derived from benzaldehyde are integral to the synthesis of fullerenes, bioactive oxacyclic compounds, dyes, and drugs, offering valuable synthetic intermediates to these respective fields. In the realm of industrial production, hexavalent chromium and other oxidizing agents are commonly employed to oxidize benzaldehyde, thereby facilitating the production of benzophenone. The significance of benzaldehyde within the pharmaceutical industry is self-evident, as it serves as a crucial raw material for the production of a diverse array of medications. This includes antihistamines, which play a pivotal role in medical treatment. Moreover, benzaldehyde finds extensive application in the production of agricultural chemicals and perfumes, testament to its wide-ranging utility and value. In the manufacture of perfumes, benzaldehyde is utilized as a fixative, thereby enhancing the stability of fragrances. This enables perfumes to retain their enchanting scents for an extended duration. In polymerization reactions, benzaldehyde functions as a terminating group in chemical reactions, thereby playing a pivotal role in regulating the progression of polymerization reactions. In the synthesis of various compounds, such as modafinil, benzhexol, and diphenyline, benzaldehyde is an indispensable precursor. Particularly in the preparation of modafinil, diphenylmethanol emerges as a key intermediate, playing a decisive role in the subsequent synthesis of the drug. This further underscores the importance of benzaldehyde in the pharmaceutical industry and provides robust support for the advancement of associated industries. As a versatile organic compound, benzaldehyde boasts extensive application prospects in both industrial and medical contexts. Its value and significance cannot be overlooked, given its pivotal role in facilitating the production of a wide array of substances, ranging from pharmaceuticals and agricultural chemicals to perfumes.
Benzenemethanol, .alpha.-phenyl-: ACTIVE
Computed Properties
Molecular Weight:184.23
XLogP3:2.7
Hydrogen Bond Donor Count:1
Hydrogen Bond Acceptor Count:1
Rotatable Bond Count:2
Exact Mass:184.088815002
Monoisotopic Mass:184.088815002
Topological Polar Surface Area:20.2
Heavy Atom Count:14
Complexity:137
Covalently-Bonded Unit Count:1
Compound Is Canonicalized:Yes
Recommended Suppliers of Benzhydrol
-
![China]() CN
CN
4 YRS
Business licensed Certified factoryManufactory Supplier of Agrochemicals,Daily Chemicals,Catalysts & Chemical Auxiliary Agents,Extract,Inorganic Chemicals,Organic Intermediate,Pigment & Dyestuff,Polymer,Flavour & Fragrance,Basic Organic Chemicals,Pharmaceutical -
![China]() CN
CN
2 YRS
Business licensed Certified factoryManufactory Supplier of N-Diethyl-m-toluamide,6-Methyluracil,Anisole,4-Dimethoxybenzoic acid,CS (lacrimator),Daucosterol,N,N-Formylmorpholine,Epichlorohydrin,Tetramethylguanidine,Dimethicone,3 -
![China]() CN
CN
3 YRS
Business licensed Certified factoryManufactory Supplier of chemicalInquiryUnit Price: $80-90 /KG FOBCAS No.: 91-01-0Grade: Industrial GradeContent: 99%
Learn More Other Chemicals
-
Celery ketone
3720-16-9
-
Cetalox
3738-00-9
-
(-)-Ambrox
6790-58-5
-
1-Methoxy-1-methylcyclododecane Formula
37514-30-0
-
Benzyl tiglate Formula
37526-88-8
-
2-Methyldodecanal Formula
37596-36-4
-
5-Cyclohexadecen-1-one Structure
37609-25-9
-
2-Undecen-1-ol Structure
37617-03-1
-
What is 2-Methyl-3-pentenoic acid
37674-63-8
-
What is Myrac aldehyde
37677-14-8





