6-Bromo-2-naphthalenol
-
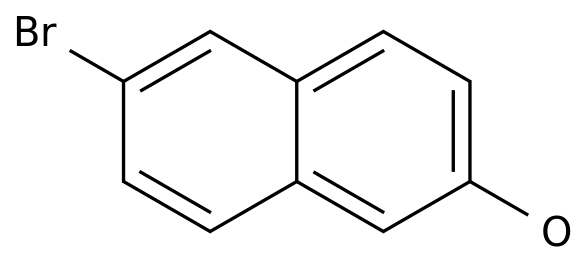
6-Bromo-2-naphthalenol
structure -
-
CAS No:
15231-91-1
-
Formula:
C10H7BrO
-
Chemical Name:
6-Bromo-2-naphthalenol
-
Synonyms:
2-Naphthalenol,6-bromo-;2-Naphthol,6-bromo-;6-Bromo-2-naphthalenol;6-Bromo-β-naphthol;6-Bromo-2-naphthol;2-Bromo-6-hydroxynaphthalene;6-Bromo-2-hydroxynaphthalene;2-Hydroxy-6-bromonaphthalene;6-Hydroxy-2-bromonaphthalene;2-Bromo-6-naphthol;6-Bromo-2-naphthalinol;NSC 17563
- Categories:
-
CAS No:
Description
off-white to beige powderChEBI: A member of the class of naphthols that is 2-naphthol carrying an additional bromo substituent at position 6.
6-bromo-2-naphthol is a member of the class of naphthols that is 2-naphthol carrying an additional bromo substituent at position 6. It is a member of naphthols and an organobromine compound. It derives from a 2-naphthol.
6-Bromo-2-naphthalenol Basic Attributes
223.07
223.07
1100270
239-279-0
87F10AHZ3O
17563
DTXSID5074551
29037990
Characteristics
20.2
3.4
Off-white to beige Powder
1.4412 (rough estimate)
127-128 °C
130°C (rough estimate)
167.8±20.4 °C
1.5700 (estimate)
Store at RT.
1.71E-05mmHg at 25°C
Safety Information
NONH for all modes of transport
3
36/37/38
26-36-37/39
QL3365000
Xi
Irritant
Stable at room temperature in closed containers under normal storage and handling conditions.
P261-P305 + P351 + P338
H315-H319-H335
|Warning|H315 (100%): Causes skin irritation [Warning Skin corrosion/irritation]|P261, P264, P271, P280, P302+P352, P304+P340, P305+P351+P338, P312, P321, P332+P313, P337+P313, P362, P403+P233, P405, and P501|Aggregated GHS information provided by 48 companies from 7 notifications to the ECHA C&L Inventory. Each notification may be associated with multiple companies.
6-Bromo-2-naphthalenol Use and Manufacturing
Derived from 2-naphthol through bromination and reduction. Add 2-naphthol and glacial acetic acid to the reaction pot, and then add the solution made of bromine and acetic acid. Add bromine, add water and heat to boiling, then add moss-like tin in batches when it is cooled to 100°C, and then boil it for 3 hours after addition, add 6-bromo-2-naphthol to the appropriate amount of cold water with stirring to precipitate, wash it with water, Filter and dry at 100°C to get crude product. After vacuum distillation, recrystallized to get the finished product.
6-Bromo-2-naphthol is a flavonoid molecule that shows steroid hormone activity and may be useful on anticancer therapy.
Computed Properties
Molecular Weight:223.07
XLogP3:3.4
Hydrogen Bond Donor Count:1
Hydrogen Bond Acceptor Count:1
Exact Mass:221.96803
Monoisotopic Mass:221.96803
Topological Polar Surface Area:20.2
Heavy Atom Count:12
Complexity:160
Covalently-Bonded Unit Count:1
Compound Is Canonicalized:Yes
Recommended Suppliers of 6-Bromo-2-naphthalenol
-
![China]() CN
CN
3 YRS
Business licensed Certified factoryManufactory Supplier of Flavors & Fragrances,Catalyst & Auxiliary,Intermediates,Dyes & Pigments,Inorganic Chemistry,petro chemicals,Surfactant,Food Additives,Water Treatment Chemicals -
![China]() CN
CN
4 YRS
Business licensedTrader Supplier of Intermediates,Building blocks,API,Silicones,Peptides,Lab chemicals,Biochemicals,Pharmaceuticals,Screening Compounds,Food Additives
Learn More Other Chemicals
-
2-Bromophenacyl bromide, 90%
49851-55-0
-
1H-Indazol-7-ol
81382-46-9
-
6-Chloro-4-forMyl-nicotinic acid
1031433-06-3
-
3-Hydroxy-2,4,5-trifluorobenzoic acid Formula
116751-24-7
-
2-Methyl-1-heptene Formula
15870-10-7
-
1-(3,5-Dinitrophenyl)ethanone Formula
14401-75-3
-
α-Amino-3-bromobenzeneacetic acid Structure
79422-73-4
-
6-(BROMOMETHYL)-1,3-BENZOTHIAZOLE,97% Structure
499770-85-3
-
What is ethyl 5-hydroxy-2-Methylnicotinate
60390-47-8
-
What is 2-bromo-1-(4-bromo-3-fluorophenyl)ethanone
1003879-02-4





