1-(1,1-Dimethylethyl) 2-borono-1-pyrrolidinecarboxylate
-
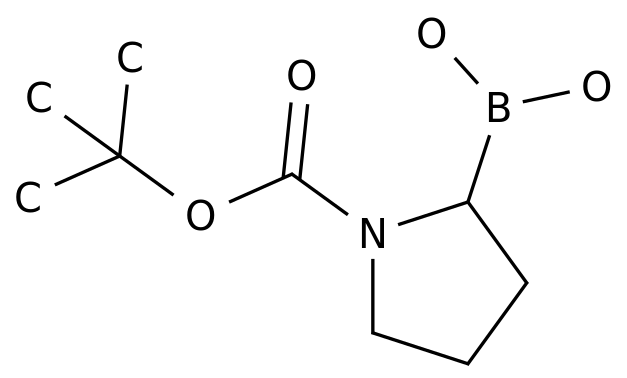
1-(1,1-Dimethylethyl) 2-borono-1-pyrrolidinecarboxylate
structure -
-
CAS No:
149682-75-7
-
Formula:
C9H18BNO4
-
Chemical Name:
1-(1,1-Dimethylethyl) 2-borono-1-pyrrolidinecarboxylate
-
Synonyms:
1-Pyrrolidinecarboxylic acid,2-borono-,1-(1,1-dimethylethyl) ester;1-(1,1-Dimethylethyl) 2-borono-1-pyrrolidinecarboxylate;[1-(tert-Butoxycarbonyl)pyrrolidin-2-yl]boronic acid;NT 0187;[1-[(2-Methylpropan-2-yl)oxycarbonyl]pyrrolidin-2-yl]boronic acid;1-N-Boc-Pyrrolidin-2-ylboronic acid;147228-19-1
- Categories:
-
CAS No:
1-(1,1-Dimethylethyl) 2-borono-1-pyrrolidinecarboxylate Basic Attributes
215.05452
215.05
604-696-2
DTXSID70376284
2933990090
Characteristics
70
0.75570
1.1±0.1 g/cm3
349.2°C at 760 mmHg
165.0±30.7 °C
1.489
2.89E-06mmHg at 25°C
1-(1,1-Dimethylethyl) 2-borono-1-pyrrolidinecarboxylate Use and Manufacturing
Synthesis of (2R)-boroPro- (lS, 2S, 3R, 5S)-pinanediol ester, hydrochloride (2); [0280] A flame dried round bottom flask equipped with a magnetic stir bar was charged with N-Boc-pyrrolidine (20 g, 117 mmol, 1 eq) and dry THF (60 mL) under a nitrogen atmosphere. The clear colorless solution was cooled to-78°C and a solution of s- BuLi (100 mL of a 1.4 M solution in cyclohexane, 140 mmol) was added slowly over a 30 minute period. The light orange colored solution was stirred at-78°C for 3 hours followed by treatment with B (OMe) 3 (39 mL, 350 mmol) after which the cooling bath was removed and the clear colorless solution slowly warmed to 0°C. Upon reaching 0°C, the reaction was quenched with a small amount of water (-2 mL), allowed to warm to room temp then extracted into 2 N NaOH (250 mL) and backwashed with additional EtOAc (150 mL). The aqueous phase was acidified to pH 3 by the addition of 2 N HCl and then extracted with EtOAc (3 x 120 mL). The organic extracts were combined and dried over Na2SO4 and concentrated to produce the free boronic acid (22.08 g, 103 mmol) as a sticky white solid in 88percent yield. Without further purification the boronic acid was dissolved in tert-butyl methyl ether (150 mL) and with constant stirring (+) -pinanediol (17.5 g, 103 mmol) was added at room temperature. After 18 hr the ether was removed and the (+) -pinanediol boronic ester was purified by column chromatography (silica gel, 1: 3 hexanes/EtOAc) to give a clear thick oil (26.84 g, 76.8 mmol, 76percent yield, Rf= 0.6 using a 2: 1 hexane/ethyl acetate eluant, made visual via 12 and/or PMA stain). Removal of the Boc protecting group was achieved by dissolving the oil in dry ether, cooling to 0°C in an ice bath and with constant stirring dry HCl (g) was bubbled into the solution for 10 minutes. After 2 hours a white precipitate developed in the flask and the ether and excess HCl were removed in vacuo to afford the racemic HCl salt as a white solid. Crystallization and isolation of the desired isomer was performed by dissolving the HCI salt in a minimal amount of dichloromethane (250 mL) with gentle heating to facilitate a homogenous solution followed by continuous stirring for 8 hours to yield a fluffy white precipitate that was collected by vacuum filtration, dried and then dissolved in minimal 2-propanol (-200 mL) with gentle heating until homogenous. The alcoholic solution was stirred over night and the resulting white precipitate was collected by vacuum filtration affording isomerically pure 1 as a white solid. (7.0 g, 27 mmol, 23percent yield).'H NMR (400 MHz, D20) 8 4.28 (d, J= 8.0 Hz, lH), 3.06 (m, 3H), 2.18 (m, 1H), 1.96 (m, 2H), 1.78 (m, 3 H), 1.62 (m, 2H), 1.21 (s, 3H), 1.05 (m, 5H), 0.84 (d, J=12 Hz, 2H), 0.71 (s, 2H), 0.62 (s, 3H).A flame dried round bottom flask equipped with a magnetic stir bar was charged with N-Boc-pyrrolidine (10g, 58mmol, 1eq) and dry THF (40mL) under a nitrogen atmosphere. The clear colorless solution was cooled to −78°C and a solution of s-BuLi (64mL of a 1.0M solution in cyclohexane, 64mmol) was added slowly over a 30min period. The light orange colored solution was stirred at −78°C for 3h followed by treatment with B(OMe)
A boc-protected cyclic amine used in the preparation of heterocyclic boronic acid compounds.
Computed Properties
Molecular Weight:215.06
Hydrogen Bond Donor Count:2
Hydrogen Bond Acceptor Count:4
Rotatable Bond Count:3
Exact Mass:215.1328882
Monoisotopic Mass:215.1328882
Topological Polar Surface Area:70
Heavy Atom Count:15
Complexity:239
Undefined Atom Stereocenter Count:1
Covalently-Bonded Unit Count:1
Compound Is Canonicalized:Yes
Learn More Other Chemicals
-
1-(1,1-Dimethylethyl) 4-(2-chloro-4-pyrimidinyl)-1,3-piperazinedicarboxylate
1261229-52-0
-
1-(1,1-Dimethylethyl) 4-methyl N-[(1,1-dimethylethoxy)carbonyl]-L-aspartate
34582-31-5
-
1-(1,1-Dimethylethyl) 4-[(3-carboxyphenyl)sulfonyl]-1-piperazinecarboxylate
874838-79-6
-
1-(1,1-Dimethylethyl) 7-(2-carboxyethenyl)-2,3-dihydro-1H-indole-1-carboxylate Formula
692287-84-6
-
1-(1,1-Dimethylethyl) 3-[[(carboxymethyl)ethylamino]methyl]-1-pyrrolidinecarboxylate Formula
1353945-75-1
-
1-(1,1-Dimethylethyl) 2,3-dihydro-2-(2-propen-1-yl)-1H-benzimidazole-1,2-dicarboxylate Formula
1255574-64-1
-
1-(1,1-Dimethylethyl) 4-methyl 4-[[(phenylmethoxy)carbonyl]amino]-1,4-piperidinedicarboxylate Structure
392331-67-8
-
5,10-Dioxa-2,8-diazaundecanoic acid, 3-carboxy-7-[(1R)-1-(1,1-dimethylethoxy)ethyl]-11-(9H-fluoren-9-yl)-6,9-dioxo-, 1-(1,1-dimethylethyl) ester, (3S,S)- Structure
944283-12-9
-
What is 1-(1,1-Dimethylethyl) 2-borono-6-[[(1,1-dimethylethyl)dimethylsilyl]oxy]-1H-indole-1-carboxylate
848357-99-3
-
What is 1-(1,1-Dimethylethyl) (2R)-2-[[3-(trifluoromethyl)phenyl]methyl]-1,2-pyrrolidinedicarboxylate
959576-50-2
1-(1,1-Dimethylethyl) 2-borono-1-pyrrolidinecarboxylate
SDSRequest for Quotation




