6-Hydroxy-3,4-dihydro-2(1H)-quinolinone
-
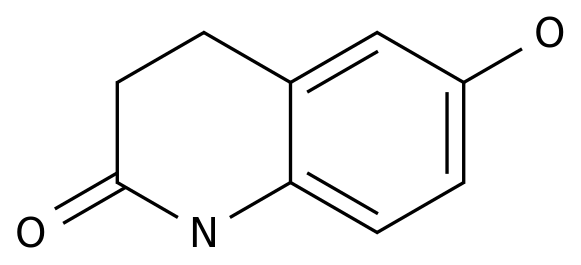
6-Hydroxy-3,4-dihydro-2(1H)-quinolinone
structure -
-
CAS No:
54197-66-9
-
Formula:
C9H9NO2
-
Chemical Name:
6-Hydroxy-3,4-dihydro-2(1H)-quinolinone
-
Synonyms:
2(1H)-Quinolinone,3,4-dihydro-6-hydroxy-;Carbostyril,3,4-dihydro-6-hydroxy-;Hydrocarbostyril,6-hydroxy-;3,4-Dihydro-6-hydroxy-2(1H)-quinolinone;6-Hydroxy-3,4-dihydrocarbostyril;6-Hydroxy-1,2,3,4-tetrahydro-2-quinolinone;3,4-Dihydro-6-hydroxycarbostyril;6-Hydroxy-2-oxo-1,2,3,4-tetrahydroquinoline;6-Hydroxy-3,4-dihydro-2(1H)-quinolinone;6-Hydroxy-3,4-dihydro-1H-quinolin-2-one;2-Oxo-1,2,3,4-tetrahydroquinolin-6-ol;3,4-Dihydroquinoline-2,6-diol
- Categories:
-
CAS No:
6-Hydroxy-3,4-dihydro-2(1H)-quinolinone Basic Attributes
163.17
163.17
611-111-4
2C5NDT39OC
DTXSID60202548
2933790090
Safety Information
NONH for all modes of transport
3
R36/37/38
S26
Xi
P305 + P351 + P338
H315-H319-H335
|Warning|H315 (100%): Causes skin irritation [Warning Skin corrosion/irritation]|P261, P264, P271, P280, P302+P352, P304+P340, P305+P351+P338, P312, P321, P332+P313, P337+P313, P362, P403+P233, P405, and P501|Aggregated GHS information provided by 43 companies from 5 notifications to the ECHA C&L Inventory. Each notification may be associated with multiple companies.
6-Hydroxy-3,4-dihydro-2(1H)-quinolinone Use and Manufacturing
2) To the 98 g of the product of the step 1), N-(4-methoxyphenyl)-3-chloroacrylamide, 500 ml of tetrahydrofuran and 98 g of palladium chloride were added.Under 5 kg pressure, Warming up to 100 to 110 ° C for 3 hours, after cooling to room temperature, Filtration and complete spin-drying to give the crude 6-hydroxy-3, 4-dihydro-2(1H)-quinolinone;3) The crude 6-hydroxy-3, 4-dihydro-2(1H)-quinolinone obtained in the step 3) was recrystallized from 50 ml of ethanol, and the activated carbon was decolorized.Filtration gave 74 g of 6-hydroxy-3, 4-dihydro-2(1H)-quinolinone as an off-white solid, yield 95percent.The total yield was 85percent.Step 2:Into a 2 L three-necked flask, 96.1 g (0.45 mol) of N- (4'-methoxyphenyl) -3-chloropropanamide and150g (2.05mol) N, N-dimethylacetamide to start stirring, To the system was added 240.0 g (1.8 mol) of aluminum trichloride, Plus Bi, warmed to 150 , incubated for 2h, TLC detection reaction was completed, stirred and cooled to 60 , To the system slowly added 1500g ice water quench, Precipitated product, filtered, and the resulting crude product was recrystallized from 1000 mL of ethanol, 63.5 g of a white-like solid was obtained, Namely 6-hydroxy-3, 4-dihydroquinolin-2-one, Yield 86.5percent (4-methoxyaniline yield 76.5percent), purity99.20percent.2) To the 98 g of the product of the step 1), N-(4-ethoxyphenyl)-3-chloroacrylamide, 500 ml of tetrahydrofuran and 98 g of palladium chloride were added.Under the pressure of 5 kg, the temperature was raised to 100 to 110 ° C for 3 hours, and after cooling to room temperature, Filtration and complete spin-drying to give the crude 6-hydroxy-3, 4-dihydro-2(1H)-quinolinone;3) The crude 6-hydroxy-3, 4-dihydro-2(1H)-quinolinone obtained in the step 3) was recrystallized from 50 ml of ethanol, and the activated carbon was decolorized.Filtration gave 74 g of 6-hydroxy-3, 4-dihydro-2(1H)-quinolinone as an off-white solid, yield 87percent.The total yield was 75percent.p-Aminophenol [0.5 gm (4.58 mmol)] was dissolved in 30 ml of each methylene chloride and saturated aqueous bicarbonate solution and stirred for 5 minutes at ambient temperature. 3-Chloropropionyl chloride [0.49 ml (5.04 mmol.)] was added over 10 min. and the reaction mixture was stirred at ambient temperature for 4 hrs. A large amount of precipitate was observed. The solids were filtered and dried to afford 0.82 gm ( 90 percent) of an off-white solid. MS APCI m/e 200 (p+1). This product (0.82 gm (4.1 mmol.)) was combined with 1.6 gm (12.3 mmol.) aluminum chloride as a mixture of solids. The mixture was then heated in an oil bath at 210 °C for 10 minutes or until the gas evolution ceases. The reaction mixture was then allowed to cool to ambient temperature and then quenched in an ice/water mixture. The aqueous phase was extracted with ethyl acetate which was separated, dried over sodium sulfate and evaporated in vacuo to a light brown solid 0.58 gm (87 percent) MS APCI m/e 164 (p+1).
A metabolite of Cilostazole.
Computed Properties
Molecular Weight:163.17
XLogP3:0.7
Hydrogen Bond Donor Count:2
Hydrogen Bond Acceptor Count:2
Exact Mass:163.063328530
Monoisotopic Mass:163.063328530
Topological Polar Surface Area:49.3
Heavy Atom Count:12
Complexity:193
Covalently-Bonded Unit Count:1
Compound Is Canonicalized:Yes
Recommended Suppliers of 6-Hydroxy-3,4-dihydro-2(1H)-quinolinone
-
![China]() CN
CN
4 YRS
Business licensedTrader Supplier of Intermediates,Building blocks,API,Silicones,Peptides,Lab chemicals,Biochemicals,Pharmaceuticals,Screening Compounds,Food Additives -
![China]() CN
CN
3 YRS
Business licensedTrader Supplier of 1H,1H,2H,2H-PERFLUORODECYLTRIMETHOXYSILANE -
![China]() CN
CN
2 YRS
Business licensedTrader Supplier of CHEMICALS,REAGENTSInquiryCAS No.: 54197-66-9Grade: Pharmacy GradeContent: 99%
Learn More Other Chemicals
-
6-Methoxy-7-[(1-methyl-4-piperidinyl)methoxy]-4(3H)-quinazolinone
264208-69-7
-
N-Carbamoylputrescine
6851-51-0
-
4-[3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]oxy-9-(3,4,5-trimethoxyphenyl)-5a,6,8a,9-tetrahydro-5H-[2]benzofuro[6,5-f][1,3]benzodioxol-8-one
11024-59-2
-
3-(Acetylamino)-5-[(acetylamino)methyl]-2,4,6-triiodobenzoic acid Formula
440-58-4
-
4-[[6-(cyclohexylmethoxy)-7H-purin-2-yl]amino]benzenesulfonamide Formula
444722-95-6
-
9-Octadecen-1-amine Formula
1838-19-3
-
(10R)-10-Hydroxyoctadecanoic acid Structure
5856-32-6
-
Undecoylium chloride Structure
1337-59-3
-
What is Phenyl methylcarbamate
1943-79-9
-
What is N,N-diethyl-2-(6-fluoro-1H-indol-3-yl)ethanamine
2836-69-3
6-Hydroxy-3,4-dihydro-2(1H)-quinolinone
SDSRequest for Quotation





