1,2-Dimethylimidazole
-
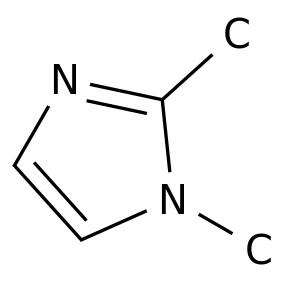
1,2-Dimethylimidazole
structure -
-
CAS No:
1739-84-0
-
Formula:
C5H8N2
-
Chemical Name:
1,2-Dimethylimidazole
-
Synonyms:
1H-Imidazole,1,2-dimethyl-;Imidazole,1,2-dimethyl-;1,2-Dimethyl-1H-imidazole;1,2-Dimethylimidazole;N,2-Dimethylimidazole;PC CAT DMI;Toyocat DP 70;SN 25;SN 25 (vulcanizing agent);Curezol 1,2DMZ;NSC 111174;Toyocat DMI;Toyocat DM 70;1,2DMZ;Nichigo Imidazole 1,2-DMI;Curezol 12DMZ;Lupragen DMI;Dabco 2040;Dabco 2039;Dabco 2041;Kaolizer 390;204854-16-0;226992-37-6;259222-76-9;263899-38-3;1821216-04-9
- Categories:
-
CAS No:
1,2-Dimethylimidazole Basic Attributes
96.133
96.13
217-101-2
Q41BC3GRJB
111174
DTXSID2061939
29332990
Characteristics
17.8
0.4
Liquid; OtherSolid
1.084 g/mL at 25 °C(lit.)
39-41 °C
206 °C
198 °F
1.5000 (estimate)
soluble in water.
Keep container closed when not in use. Store in a cool, dry, well-ventilated area away from incompatible substances. Corrosives area.
1 mm Hg ( 20 °C)
LD50 orally in Rabbit: 1300 mg/kg
Safety Information
III
8
UN 3263 8/PG 2
2
R22;R38;R41
24-26-2
NI4838854
Xn,Xi
Stable under normal temperatures and pressures.
P280-P305 + P351 + P338
H302-H315-H318
|Danger|H302: Harmful if swallowed [Warning Acute toxicity, oral]|P264, P270, P280, P301+P312, P302+P352, P305+P351+P338, P310, P321, P330, P332+P313, P362, and P501|H302 (100%): Harmful if swallowed [Warning Acute toxicity, oral]|P261, P264, P270, P272, P280, P301+P312, P302+P352, P305+P351+P338, P310, P321, P330, P332+P313, P333+P313, P362, P363, and P501|Aggregated GHS information provided by 236 companies from 8 notifications to the ECHA C&L Inventory. Each notification may be associated with multiple companies.
1,2-Dimethylimidazole Use and Manufacturing
As shown in Fig. 1, a high-pressure microreactor in which a micro mixer was incorporated at three locations was used.An aqueous solution of methylamine (0.33 mol / L) was fed from the tank 1 through the pump 1 at a flow rate of 0.68 mL / min, an aqueous solution of acetaldehyde (0.33 mol / L) At a flow rate of 63 mL / min, from the tank 3 via the pump 3AmmoniumNear water(0.33 mol / L) was flowed at a flow rate of 1.13 mL / min, an aqueous glyoxal solution (0.33 mol / L) was flowed from the tank 4 via the pump 4 at 0.56 mL / flow rate, and a reaction pressure of 30 MPa The reaction was carried out under pressure. The final flow rate was 8.0 mL / min. The temperature of the first micromixer and the reaction tube of 2.4 m is controlled in a thermostatic chamber 1 keeping the temperature at 25 ° C. The mixture and reaction of methylamine and acetaldehyde in the first micromixer and reaction tube Was done. In the next second micromixer in which the aqueous solution and ammonia water are mixed by this mixing, they are mixed in a thermostatic chamber 2 kept at a temperature of 25 ° C., and after sufficient mixing and reaction in a 0.1 m reaction tube After that, glyoxal was mixed, after which 1Sufficient reaction was carried out in a 0 m reaction tube. Next, the temperature of the reaction part was adjusted to 70 ° C., and the reaction was carried out in a 25 m reaction tube. Finally, it was cooled down to 5 ° C. with a heat exchanger, depressurized to normal pressure via a discharge pressure valve, and the aqueous solution obtained while cooling with ice water (0 ° C. to 4 ° C.) was collected. In this case, the reaction time (residence time in the reaction part) was 37 seconds. For the analysis, if necessary diluted 10 times with ice water and analyzed immediately using a gas chromatograph. The reaction tube was made of stainless steel (SUS 316) and had an inner diameter of 0.5 mm. As a result, the objective 1, 2-dimethylimidazole was obtained in a yield of 85.1percent.For measurement of the above quantitative yield, a gas chromatograph (GC 6890, hydrogen flame ionization detector) manufactured by Agilent was used and the column was HP INNOWAX manufactured by J & W Co., inner diameter 0.32 mm, film thickness 0.25 μm, length Analysis was carried out using 30 m. The yield of each product was determined by preparing a calibration curve for each compound using various concentrations of the product (1, 2-dimethylimidazole, 1-methylimidazole, 2-methylimidazole, imidazole) Concentration of each product obtained was converted from a calibration curve, and the yield of each product was calculated. The respective yields were 1, 2-dimethylimidazole 85.1percent, 1-methylimidazole 6.27percent, 2-methylimidazole 6.59percent and imidazole 2.06percent.To 500 ml of the input three-mouth flask in 128g of 2 - methyl imidazole and 100 ml of DMF, oil bath heated to 120 °C, the temperature next adds by drops 280g dimethyl carbonate, control temperature at 135 - 140 °C, dropping time control in 5 - 6h, after dropping in the 145 °C preserving 2h, thermal insulation after the completion of the, gas chromatographic analysis of 2 - methyl imidazole conversion 98percent. In 4.0KPa, 90 °C conditions under reduced pressure distillation, evaporate the solvent. The use of the rectifying device, in 4.0KPa under vacuum, collecting 100 - 110 °C of distillate, high purity can be obtained of the 1, 2 - dimethyl imidazole of the finished product, yield 85percent, gas chromatographic analysis of the content of 99.5percent.
Intermediates
urethane intermediate
500,000 - 1,000,000 lb
All other basic organic chemical manufacturing|1H-Imidazole, 1,2-dimethyl-: ACTIVE
Computed Properties
Molecular Weight:96.13
XLogP3:0.4
Hydrogen Bond Acceptor Count:1
Exact Mass:96.068748264
Monoisotopic Mass:96.068748264
Topological Polar Surface Area:17.8
Heavy Atom Count:7
Complexity:63.1
Covalently-Bonded Unit Count:1
Compound Is Canonicalized:Yes
Learn More Other Chemicals
-
Methyltris(methylisobutylketoxime)silane
37859-57-7
-
METHYL 3,4-BIS(BROMOMETHYL)BENZOATE
20896-23-5
-
2-(Methylthio)acetonitrile
35120-10-6
-
N-Iodosaccharin Formula
86340-94-5
-
5-Chloro-2-methoxy-4-methylaminobenzoic Acid Formula
61694-98-2
-
2-Chloro-3,6-difluorocinnamic acid Formula
261762-48-5
-
3-CHLORO-2-FLUORO-5-(TRIFLUOROMETHYL)PHENYLACETONITRILE Structure
261763-15-9
-
2-Chloro-4-fluoro-3-methoxy-1-methylbenzene Structure
261762-79-2
-
What is 2-Chloro-3,6-difluorobenzeneacetonitrile
261762-54-3
-
What is 3-chloro-N'-[4-(2,4-dichlorophenoxy)butanoyl]-1-benzothiophene-2-carbohydrazide
356103-84-9





