6-Hydroxy-7-methoxy-4(3H)-quinazolinone
-
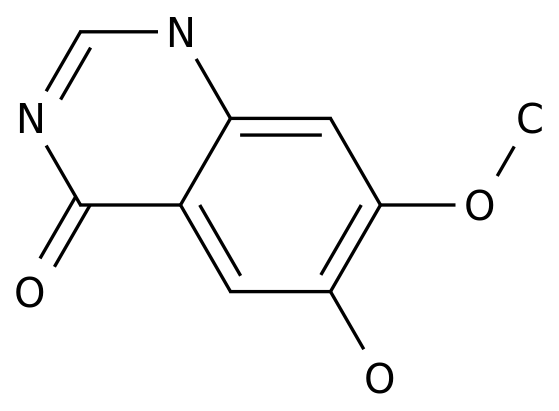
6-Hydroxy-7-methoxy-4(3H)-quinazolinone
structure -
-
CAS No:
179688-52-9
-
Formula:
C9H8N2O3
-
Chemical Name:
6-Hydroxy-7-methoxy-4(3H)-quinazolinone
-
Synonyms:
4(3H)-Quinazolinone,6-hydroxy-7-methoxy-;4(1H)-Quinazolinone,6-hydroxy-7-methoxy-;6-Hydroxy-7-methoxy-4(3H)-quinazolinone;6-Hydroxy-7-methoxy-3,4-dihydroquinazolin-4-one;7-Methoxyquinazoline-4,6-diol;6-Hydroxy-7-methoxyquinazolin-4(3H)-one
- Categories:
-
CAS No:
Characteristics
70.9
0.3
1.47±0.1 g/cm3(Predicted)
293 °C
420.0±55.0 °C(Predicted)
258.2ºC
1.659
0mmHg at 25°C
6-Hydroxy-7-methoxy-4(3H)-quinazolinone Use and Manufacturing
( 1 -2) 6-hydroxy-7-methoxyquinazolin-4(3H)-one; 3.06 g of the compound obtained in (1-1) was diluted with 20 mi of methanesulfonic acid. 2.66 g of /--methionine was added to the resulting solution and stirred at 100 The procedure for the synthesis of the title compound is depicted in Figure 1. 1-2: 6-Hydroxy-7-methoxy-3, 4-dihydroquinazolin-4-one was obtained according to W096/33980 in 93percent yield. The 6, 7-dimethoxy -3H quinazolin-4-one (5g, 0.024mol), methanesulfonic acid (30 ml) and L-methionine (5g, 0.033mol) adding 50 ml reaction flask, slow heating to 120 °C, reaction 6h, proper amount of ice added in the reaction solution, then using 40percent NaOH to pH=7, generating a large amount of white precipitation, filtration, ice water washing, get white solid, yield 90percentInto a reaction flask, concentrated sulfuric acid (607.2g; 25.5 m.eq), 6, 7-dimethoxy- quinazoline-4(3H)-one of formula-XI (50g; 1.0 m.eq) and DL-Methionine (41.5g; 1.15 m.eq) were sequentially added under stirring. The reaction mass was heated to 90-95°C and maintained for about 6h. The progress of the reaction was monitored by TLC. After completion of the reaction, cooled the reaction mass to 25-35°C and quenched by adding ice-flakes (~1300g) at 25-35°C and stirred for lh. The resulting product was filtered and suspended the wet product in DM water (800 mL). The pH of the reaction mixture was adjusted to about 7.5 with lye solution. The product was filtered, washed with DM water and dried at 70-75°C to afford 6-hydroxy-7- methoxy-quinazoline-4(3H)-one of formula-XIV (41. Og; 90.0percent by theory).A mixture of compound 2 (9.11 g, 44.3 mmol), L-methionine (7.25 g, 48.6 mmol) and methanesulfonic acid (65 mL) was heated to 120 °C for 18 h. The reaction mixture was cooled and poured into ice-water, then neutralized with 40percent NaOH to pH 7. The resultant precipitate was filtered, washed with water and dried under vacuum.The crude product was recrystallized from MeOH to afford 3 (5.06 g, 60percent) as a pale yellow solid, mp 306 °C (dec.). A suspension of 6, 7-dimethoxyquinazolin-4(3H)-one (6.18 g), methionine (4.70 g) and methanesulfonic acid (40 mL) was stirred at 130° C. for 3 h, then poured into ice-water. The reaction mixture was adjusted to pH 7 with 40percent sodium hydroxide. The mixture was filtered to give the title compound (7.10 g).General procedure: To a solution of 6, 7-dimethoxyquinazoline 1a (200.0 mg, 1.05 mmol) in ethanol (20 mL) were added 40percent peracetic acid (1.0mL, 5.26 mmol) and 0.01 mL sulfuric acid (1.8 mmol). After the reaction was stirred at 60°C for 4–12 h (see Table 2) and then cooled to room temperature, excess sodium bisulfite (541.8 mg, 5.26 mmol) was added to get rid of the peroxide. The solid was filtered off after stirring for 20 min, and the filtrate was concentrated under reduced pressure to give the crude product, which was washed by ethanol and petroleum ether to give 2a as a light yellow solid (179.7 mg, 83percent), mp>300°C.[00692] IA Step 4: A mixture of methyl 2-amino-5-hydroxy-4- methoxybenzoate (3.1 g, 15.7 mmol) and AcOH (7.1 mL) in formamide (15.5 mL) was heated at 140 °C overnight. To it was added water (20 mL) and filtered to afford 6-hydroxy-7-methoxyquinazoline-4(3//)-one as a solid (2.7 g, 89percent). Step 4. Step 4.
Recommended Suppliers of 6-Hydroxy-7-methoxy-4(3H)-quinazolinone
-
![China]() CN
CN
4 YRS
Business licensedTrader Supplier of Intermediates,Building blocks,API,Silicones,Peptides,Lab chemicals,Biochemicals,Pharmaceuticals,Screening Compounds,Food Additives -
![China]() CN
CN
4 YRS
Business licensed Certified factoryManufactory Supplier of fine chemicals,pharmaceutical materialsInquiryCAS No.: 179688-52-9Grade: industrial GradeContent: 98%
Learn More Other Chemicals
-
5-Bromo-2-methoxythiazole
446287-05-4
-
4-[(6,7-Dimethoxyquinolin-4-yl)oxy]aniline
190728-25-7
-
N-[3-Fluoro-4-[(methylamino)carbonyl]phenyl]-2-met
1332524-01-2
-
Glycine Formula
56-40-6
-
Sodium iodide Formula
7681-82-5
-
Bromo-2-pyridinylzinc Formula
218777-23-2
-
ethyl 2-oxo-2-(2-oxopyrrolidin-3-yl)acetate Structure
90090-56-5
-
2-Butyl-3-(4-(3-(dibutylamino)propoxy)benzoyl)-5-nitrobenzofuran Structure
141645-23-0
-
What is Macluraxanthone
5848-14-6
-
What is 2-(2-chlorophenyl)-5-[3-[5-(2-chlorophenyl)-1,3,4-thiadiazol-2-yl]phenyl]-1,3,4-thiadiazole
90032-82-9
6-Hydroxy-7-methoxy-4(3H)-quinazolinone
SDSRequest for Quotation





